
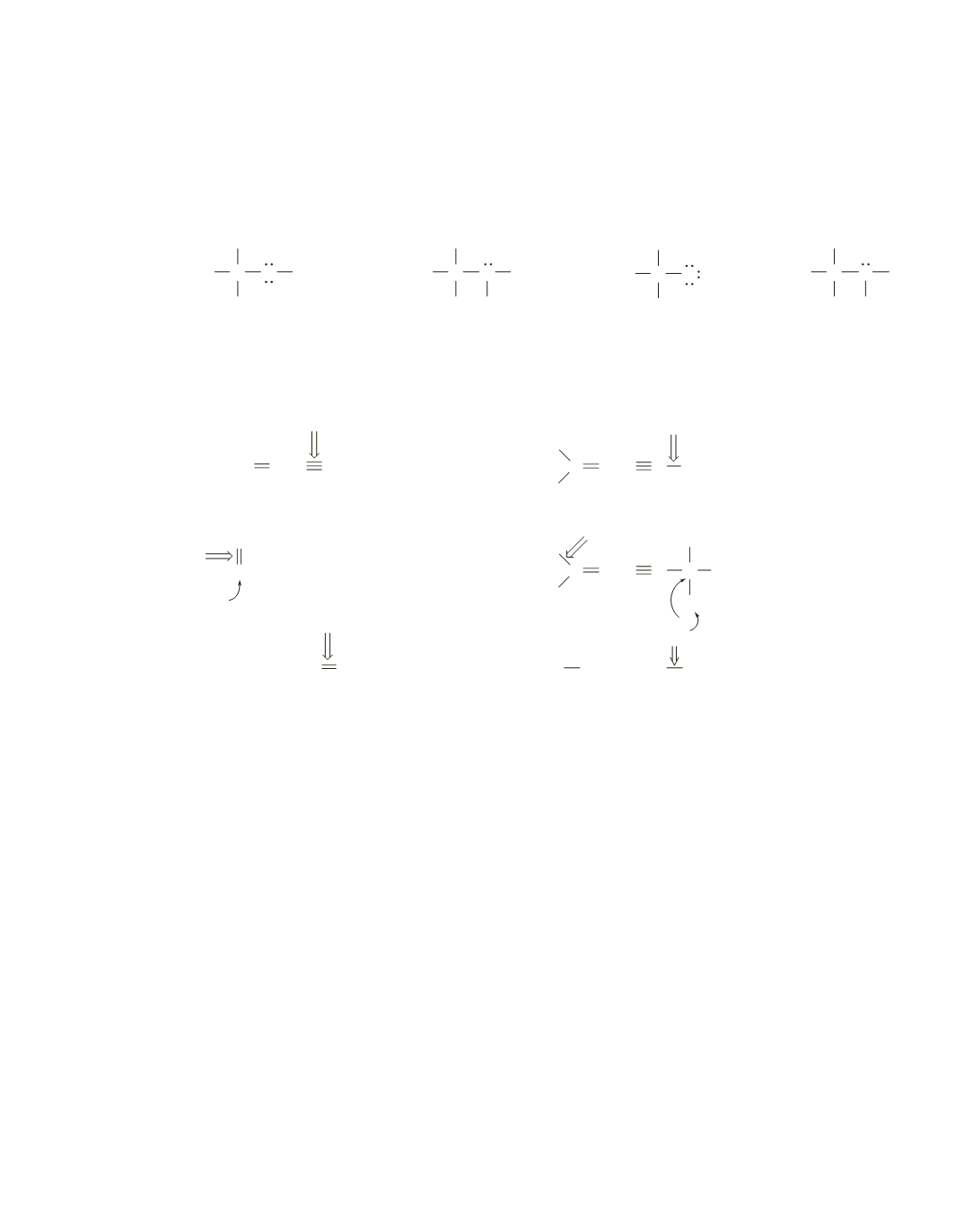
Chapter 1
87
Copyright © 2017 Pearson Education, Inc.
63.
formal charge
=
number of valence electrons
-
(number of lone-pair electrons
+
the number of bonds)
In all four compounds, H has a single bond and is neutral and each C has four bonds and is neutral.
Thus, the indicated formal charge is for O or N.
a.
H C
H
H
O H
formal charge
6
−
(4
+
2)
=
0
b.
+
H C
H
H
O H
H
formal charge
6
−
(2
+
3)
= +
1
c.
−
H C
H
H
O
formal charge
6
−
(6
+
1)
= −
1
d.
H C
H
H
N H
H
formal charge
5
−
(2
+
3)
=
0
64.
The open arrow in the structures points to the shorter of the two indicated bonds in each compound.
1.
CH
3
CH
sp
3
CHC CH
sp sp
sp
2
sp
2
2.
sp
3
CH
3
CCH
2
OH
O
sp
2
sp
2
sp
3
sp
3
3.
CH
3
NHCH
2
CH
2
N
sp
3
sp
2
CHCH
3
sp
2
sp
3
sp
3
sp
3
sp
3
4.
C CHC C
sp
sp
2
H
H
H
sp
2
sp
5.
C CHC C
sp
2
C
H
H
sp
2
sp sp
CH
3
CH
3
H
sp
3
sp
3
6.
Br
Cl
CH
2
CH
2
CH
2
sp
3
sp
3
sp
3
For
1
,
2
, and
3
:
A triple bond is shorter than a double bond, which is shorter than a single bond.
For
4
and
5
:
The greater the
s
character in the hybrid orbital, the shorter the bond formed
using that orbital, because an
s
orbital is closer than a
p
orbital to the nucleus.
Therefore, the bond formed by a hydrogen and an
sp
carbon is shorter than the bond
formed by a hydrogen and an
sp
2
carbon, which is shorter than the bond formed by a
hydrogen and an
sp
3
carbon. (See Table 1.7 on page 42 of the text.)
For
6
:
Cl forms a bond using a
3
sp
3
orbital, and Br forms a bond using a
4
sp
3
orbital.
Therefore, the
C
¬
Cl
bond is shorter.
















