
80
Chapter 1
Copyright © 2017 Pearson Education, Inc.
23.
a.
Cl
b.
O
O
c.
O
N
d.
N
24.
c.
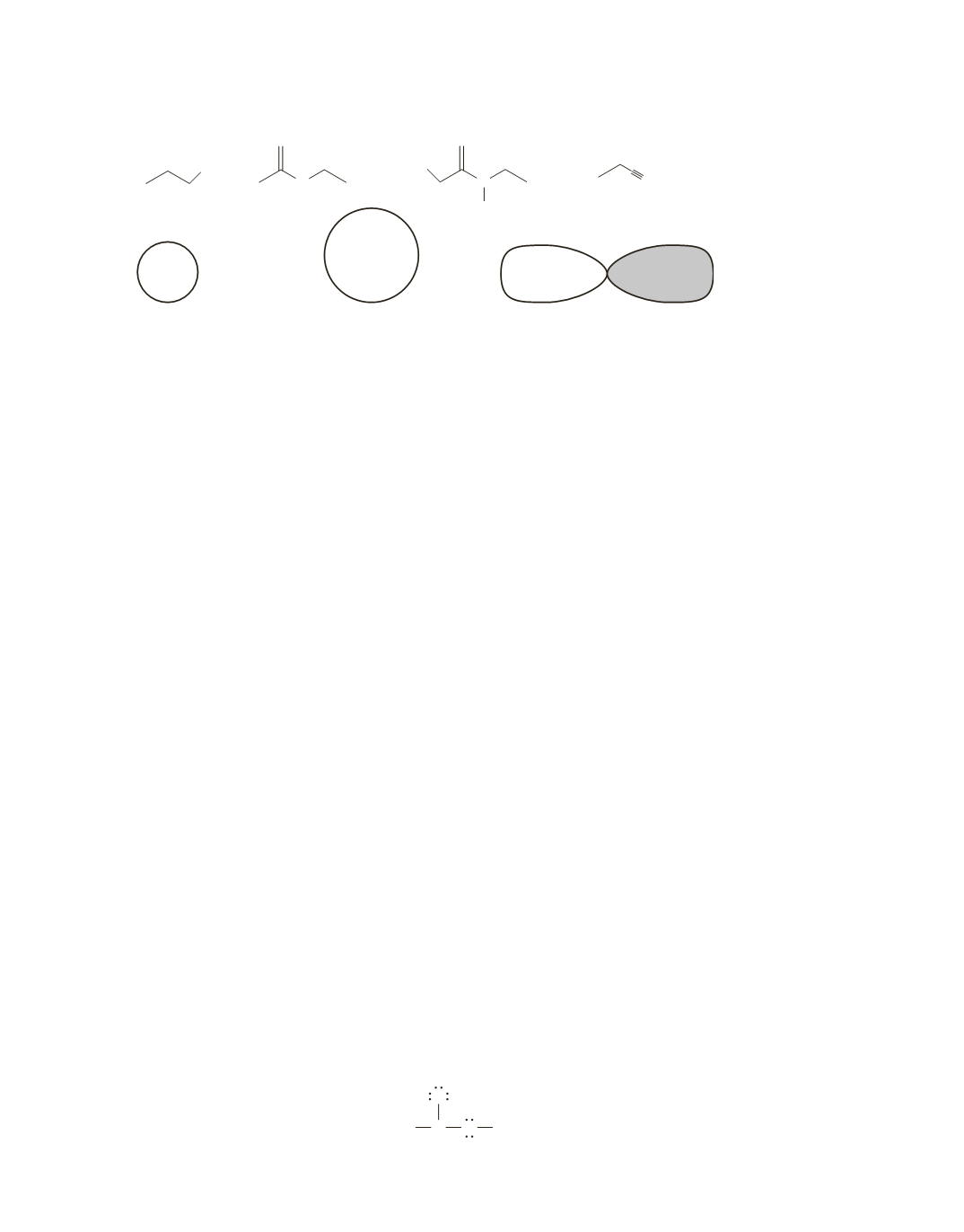
b.
a ball larger than the
ball that represents
a 3
s
orbital
a.
a ball larger than
the ball that represents
a 2
s
orbital
25.
He
2
+
has three electrons. Using Figure 1.3 on page 23 of the text, two electrons will be in a bonding molec-
ular orbital and one electron will be in an antibonding molecular orbital. Because there are more electrons
in the bonding molecular orbital than in the antibonding molecular orbital,
He
2
+
exists.
26.
a.
p
*
This involves out-of-phase interaction of atomic orbitals (the interacting orbitals have different
colors), leading to an antibonding molecular orbital. Because this example involves the side-to-
side overlap of
p
orbitals, it is a
p
*
antibonding molecular orbital.
b.
p
This involves in-phase overlap of atomic orbitals (the overlapping orbitals have the same color),
leading to a bonding molecular orbital. Because this example involves the side-to-side overlap of
p
orbitals, it is a
p
bonding molecular orbital.
c.
s
*
This involves out-of-phase interaction of atomic orbitals (the interacting orbitals have different
colors), leading to an antibonding molecular orbital. Because this example involves the end-on
overlap of atomic orbitals, it is a
s
*
antibonding molecular orbital.
d.
s
This involves in-phase overlap of atomic orbitals (the overlapping orbitals have the same color),
leading to a bonding molecular orbital. Because this example involves the end-on overlap of
atomic orbitals, it is a
s
bonding molecular orbital.
27.
The 3 carbon–carbon bonds form as a result of
sp
3
9
sp
3
overlap.
The 7 carbon–hydrogen bonds form as a result of
sp
3
9
s
overlap.
28.
The electron density of the large lobe of an
sp
3
orbital (the lobe that overlaps the
s
orbital) is greater than
the electron density of a lobe of a
p
orbital. Therefore, the overlap of an
s
orbital with an
sp
3
orbital forms
a stronger bond than does the overlap of an
s
orbital with a
p
orbital.
29.
Solved in the text.
30.
a. One
s
orbital and
three
p
orbitals form
four
sp
3
orbitals.
b. One
s
orbital and
two
p
orbitals form
three
sp
2
orbitals.
c. One
s
orbital and
one
p
orbital form
two
sp
orbitals.
31.
a(1).
Solved in the text.
b(1).
Solved in the text.
32.
a(1).
The first attempt at drawing a Lewis structure results in a carbon that does not have a complete octet
and does not form the needed number of bonds.
H C
O
O H
















