
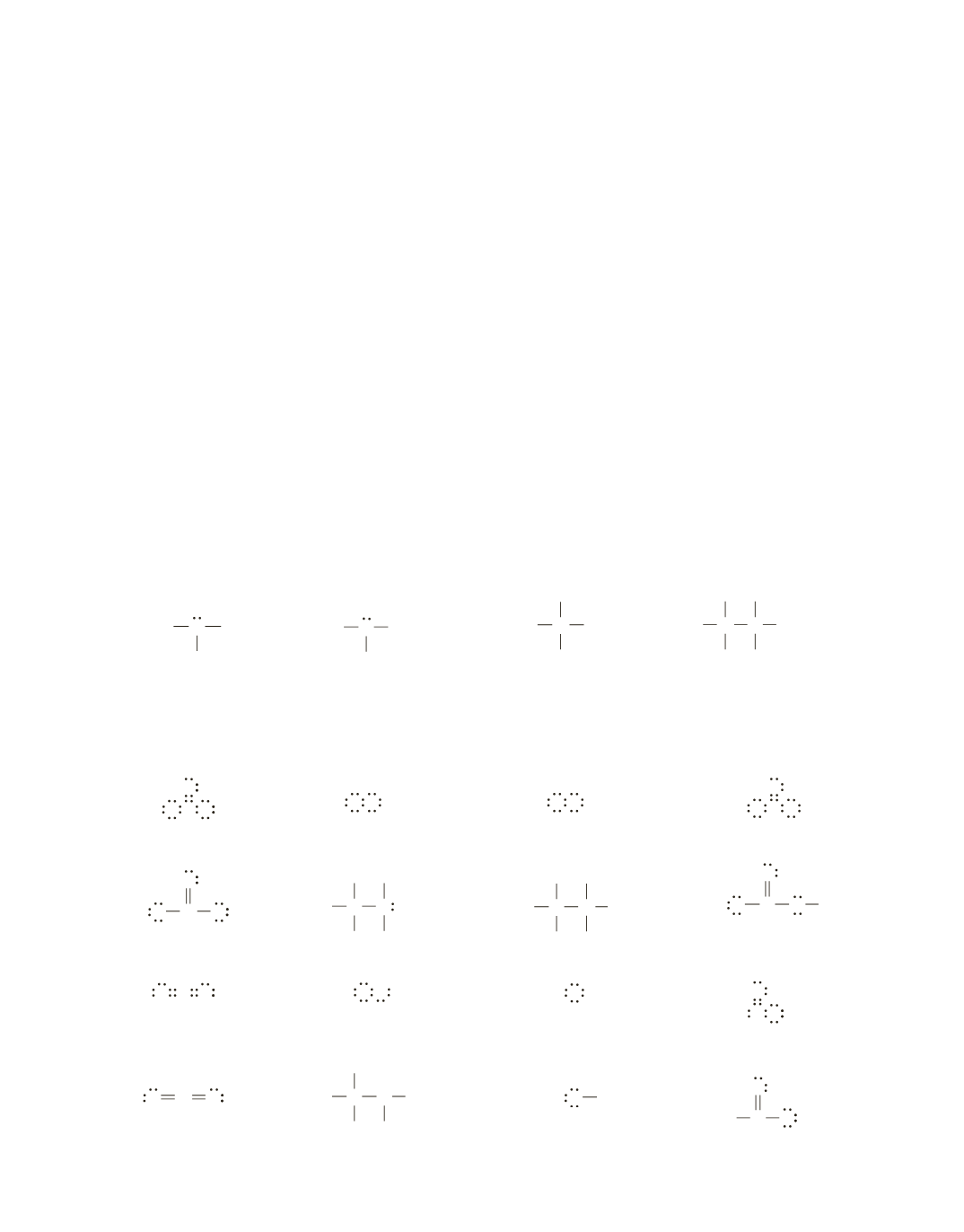
78
Chapter 1
Copyright © 2017 Pearson Education, Inc.
14.
a.
LiH and HF are polar (they have a red end and a blue end).
b.
A potential map marks the edges of the molecule’s electron cloud. The electron cloud is largest around
the hydrogen in LiH, because that hydrogen has more electrons around it than do the hydrogens in the
other molecules.
c.
Because the hydrogen of HF is blue, we know that this compound has the most positively charged
hydrogen and, therefore, will be most apt to attract a negatively charged species.
15.
By answering this question, you will see that a formal charge is a bookkeeping device. It does
not necessarily
tell you which atom has the greatest electron density or is the most electron deficient.
a.
oxygen
b.
oxygen (it is more red)
c.
oxygen
d.
hydrogen (it is the deepest blue)
Notice that in the hydroxide ion, the atom with the formal negative charge
is
the atom with the greater
electron density. In the hydronium ion, however, the atom with the formal positive charge
is not
the most
electron-deficient atom.
16.
formal charge
=
number of valence electrons
-
(number of lone-pair electrons
+
the number of bonds)
In all four structures, every H is singly bonded and thus has a formal
charge
=
1
-
(0
+
1)
=
0.
Similarly, all
CH
3
carbon atoms have four bonds and a formal
charge
=
4
-
(0
+
4)
=
0.
The formal charges on the remaining atoms:
a.
CH
3
O
H
CH
3
+
formal charge on O
6
-
(2
+
3)
=
+
1
b.
H C
H
H
−
formal charge on C
4
-
(2
+
3)
=
-
1
c.
N CH
3
CH
3
CH
3
CH
3
+
formal charge on N
5
-
(0
+
4)
=
+
1
d.
N B
H
H
−
H
H
H
H
+
formal charge on
N:
5
-
(0
+
4)
=
+
1
B:
3
-
(0
+
4)
=
-
1
17.
The bond between two atoms can be shown by a pair of dots or by a line, so there are two ways each of the
answers can be written. Remember that all lone pairs have to be shown.
a.
O
+
or
N O
−
O
O N O
−
O
−
−
+
b.
or
N
O N O
O O
+
+
c.
H C
H
C
H
H H
−
or
C C
H
−
H
H
H
H
d.
H C
H
C H
H
H
+
or
C C
H
H
+
H
H
H
e.
H C
H
N H
H
H H
+
or
C N
H
H
+
H
H
H
H
f.
Na
+
O H
Na
+
O H
−
−
or
g.
O C O
or
C O H
−
O
O
H
−
O
h.
or
H C
O
CH
O
O
−
O
−
















