
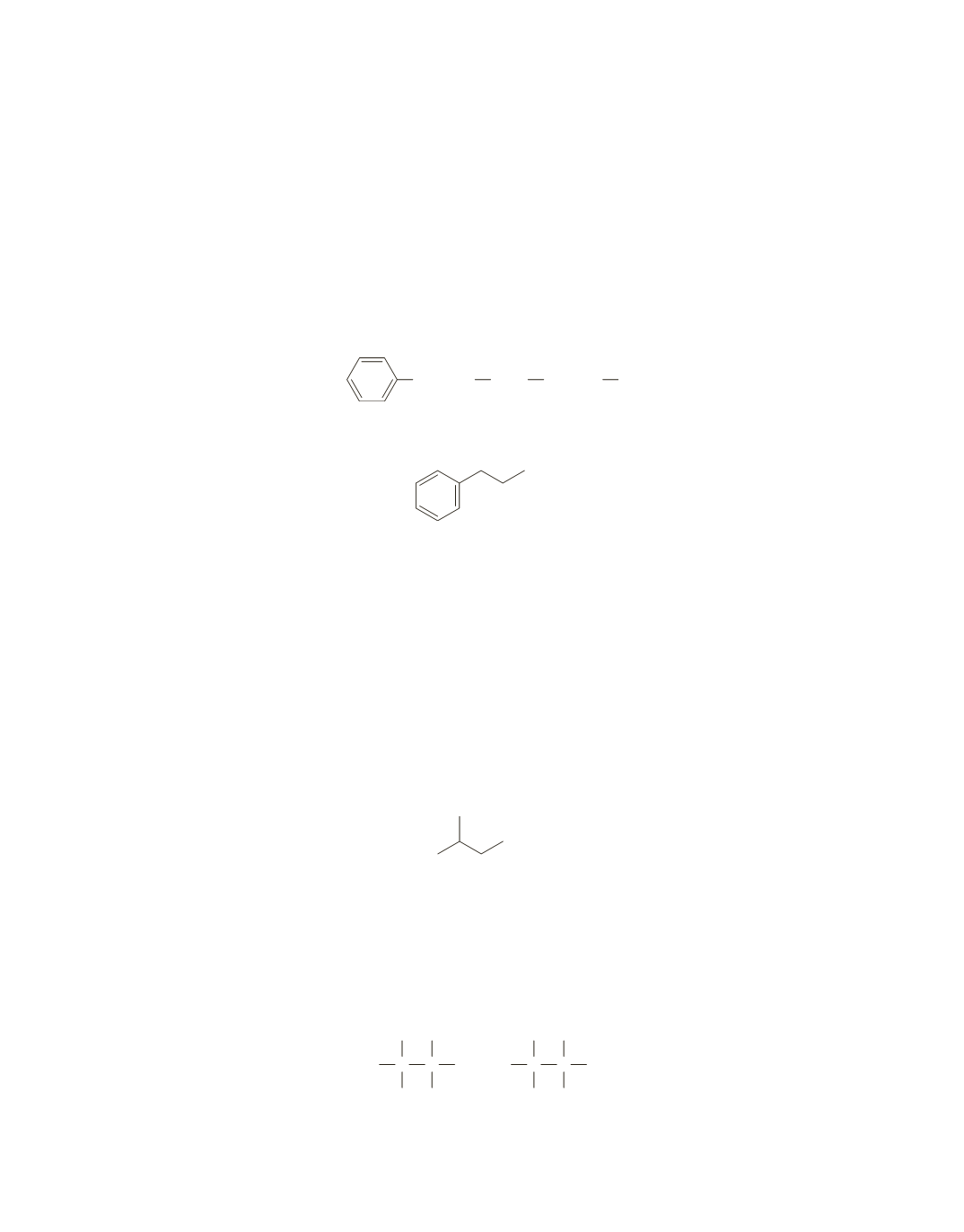
60
Spectroscopy Problems
Copyright © 2017 Pearson Education, Inc.
37.
The only signal that integrates to 1 proton is the singlet at 2.2 ppm. This must be due to the OH group of
the alcohol.
The signals centered around 7.3 ppm are given by benzene-ring protons. Because they integrate to
5 protons, the benzene ring must be monosubstituted.
The two triplets (2.8 and 3.8 ppm) that each integrate to 2 protons suggest two adjacent methylene groups.
Both signals are fairly deshielded, indicating an electronegative atom nearby.
The fragments identified at this point are a monosubstituted benzene ring, an OH group, and two adjacent
methylene groups.
HO
CH
2
CH
2
No other signals are in the NMR spectrum, so the compound must be the one shown here.
OH
38.
The doublet (0.9 ppm) that integrates to 6 protons and the multiplet (1.8 ppm) that integrates to 1 proton
suggest the presence of an isopropyl group.
Because we are told that the compound is an alcohol, the other signal that integrates to 1 proton (the triplet
at 2.4 ppm) must be due to the OH proton. The fact that the signal is a triplet indicates that the OH group is
probably attached to a methylene group.
The signal for the methylene group must be the remaining signal at 3.4 ppm because it integrates to
2 protons. The relatively high-frequency chemical shift confirms that the methylene group is attached to
the oxygen.
Putting together the isopropyl group and the methylene group that is attached to an OH group identifies the
compound as
2-methyl-1-propanol
.
OH
39.
The protons that are responsible for the doublet (1.2 ppm) that integrates to 6 protons must be adjacent to
a carbon that is attached to only one proton. Because the spectrum does not have a signal that integrates to
one proton, the compound must have two methyl groups in the same environment. Each methyl group must
be adjacent to a carbon that is attached to one proton, and those two single protons must be in identical
environments.
C
H
C H
H
H
C
H
H
H C
H
















