
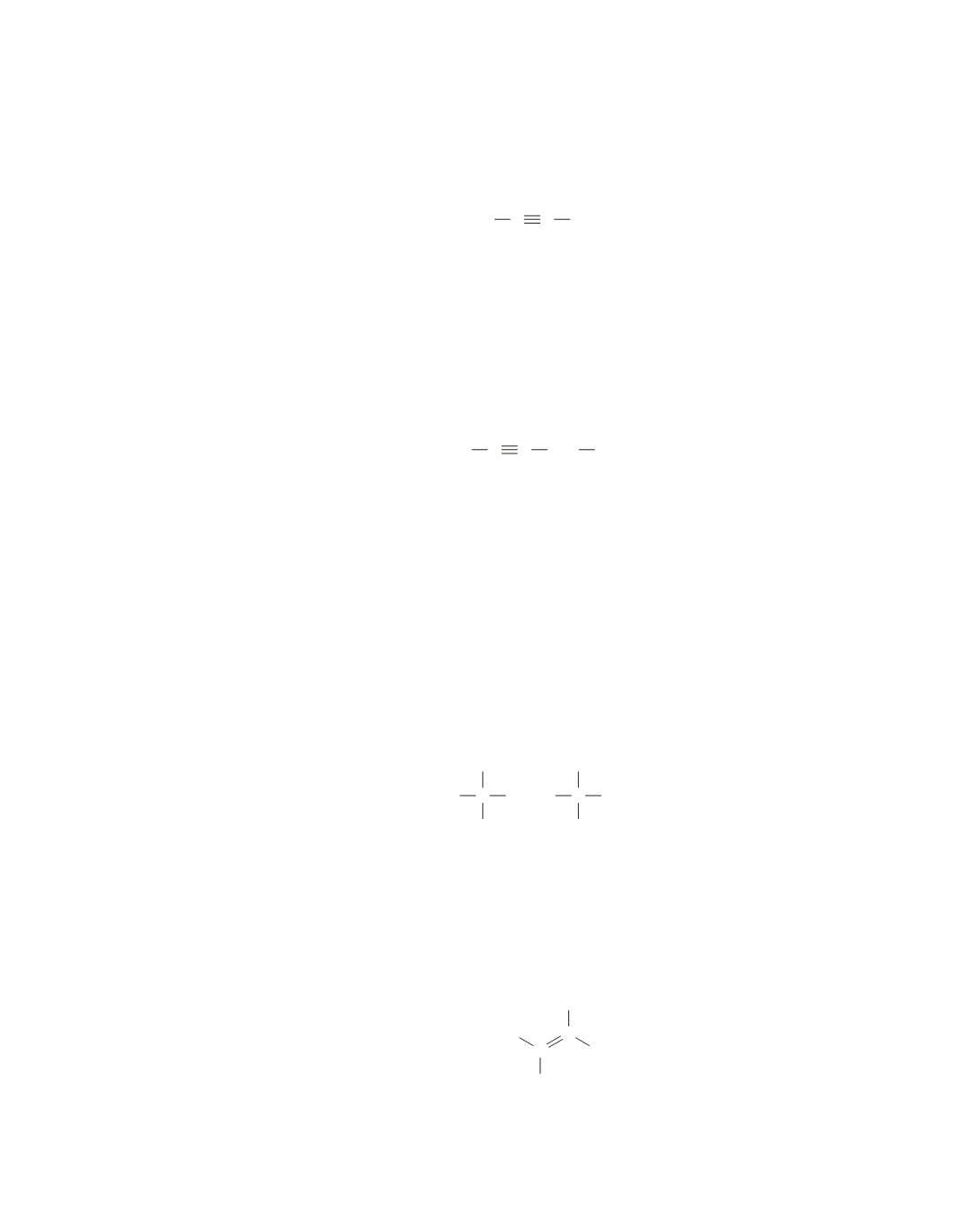
Spectroscopy Problems
59
Copyright © 2017 Pearson Education, Inc.
35.
A medium-intensity absorption at
2120 cm
-
1
indicates the presence of a carbon–carbon triple bond. The
sharp absorption at
3300 cm
-
1
is due to the
C
¬
H
stretch of a hydrogen bonded to an
sp
carbon. Thus, the
compound is a terminal alkyne.
CC H
The intense and broad peak centered at
3300 cm
-
1
is evidence of an
O
¬
H
group.
The NMR spectrum can be used to determine the connectivity between the groups. The signal (2.5 ppm)
that integrates to 1 proton is due to the proton of the terminal alkyne.
The singlet (3.2 ppm) that integrates to 1 proton must be due to the proton of the OH group.
The singlet (4.2 ppm) that integrates to 2 protons must be due to a methylene group that connects the triply
bonded carbon to the OH group. The compound is
2-propyn-1-ol
.
CCH
CH
2
OH
This arrangement explains the absence of any splitting and the highly deshielded nature of the signal for
the methylene group.
36.
The two singlets (4.9 and 5.1 ppm) that each integrate to 1 proton are vinylic protons. Therefore, we know
that the compound is an alkene.
The singlet (2.8 ppm) that integrates to 3 protons is a methyl group. The deshielding results from its being
attached to an
sp
2
carbon.
If we subtract the two vinylic protons, the two
sp
2
carbons of the alkene, and the methyl group from the
molecular formula, we are left with
CH
2
Cl.
Thus, a chloromethyl group is the fourth substituent of the
alkene and gives the singlet (4.9 ppm) that integrates to 2 protons. Its deshielding is due to the proximity to
the electronegative chlorine.
C
H
H
H
H
C Cl
H
Now we need to determine the substitution pattern of the alkene. The absence of splitting indicates that the
two vinylic protons must be attached to the same carbon. If these protons were cis or trans to each other,
they would give doublets with significant
J
values. Geminal protons attached to
sp
2
carbons have very
small
J
values, so splitting is typically not observed. (See Table 14.2 on page 644 of the text.)
Thus, the compound is
3-chloro-2-methyl-1-propene
.
ClCH
2
CH
3
C H
C
H
















