
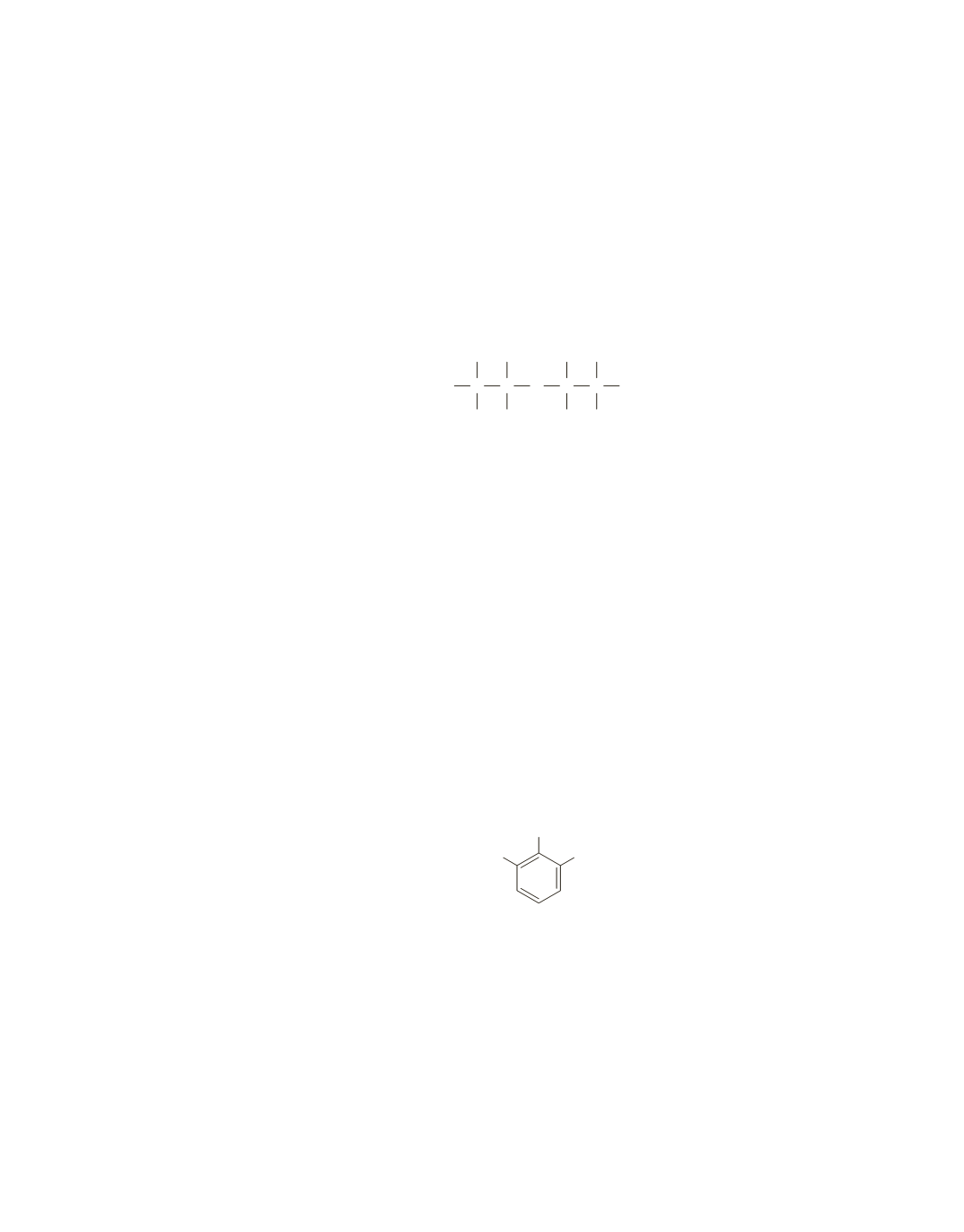
Spectroscopy Problems
63
Copyright © 2017 Pearson Education, Inc.
The triplet (1.2 ppm) that integrates to 3 protons and the quartet (3.5 ppm) that integrates to 2 protons suggests
an ethyl group. The high-frequency chemical shift of the ethyl’s methylene group and the fact that it shows
splitting only by the three protons of the methyl group indicate that the ethyl group is next to the oxygen.
The two remaining signals are both triplets (3.4 and 3.5 ppm), and each integrates to 2 protons. Thus, the
signals are due to two adjacent methylene groups. Because both signals occur at high frequencies, both
must be attached to electron-withdrawing atoms.
Because the molecular formula tells us that the compound contains a bromine, we can conclude that the
compound is
2-bromoethyl ethyl ether
.
C
H H
H
H C O
H
H
C C Br
H
HH
44.
The IR spectrum shows a strong and broad absorption at
3300 cm
-
1
,
indicating that the compound is
an alcohol.
The signals in the NMR spectrum between 6 and 7 ppm indicate a benzene ring. Because these signals
integrate to a total of 3 protons, the benzene ring must be trisubstituted.
Because the signal at 6.3 ppm is a doublet, it must be adjacent to one proton, and because the signal at 6.7 ppm is
a triplet, it must be adjacent to two protons. Thus, the three benzene-ring protons must be adjacent to one other.
The singlet at 8.7 ppm is the only signal in the spectrum that can be attributed to the proton of the OH group.
Because the signal integrates to 2 protons, the compound must have two OH groups in the same environment.
The singlet (2.0 ppm) that integrates to three protons indicates that the compound has a methyl group that
is not adjacent to a carbon that is attached to any hydrogens.
Therefore, we know that the three substituents that are attached to adjacent carbons on the benzene ring are
two OH groups and a methyl group. Because the OH groups are in the same environment, the compound
must be the one shown here.
CH
3
HO
OH
45.
We are told that the compound is an alcohol. Because the singlet (1.4 ppm) is the only signal that integrates
to 1 proton, it must be the signal given by the OH group.
We know that a triplet that integrates to 3 protons and a quartet that integrates to 2 protons are characteristic
of an ethyl group. In this case, the triplet (0.8 ppm) integrates to 6 protons and the quartet integrates to
4 protons. Therefore, the compound must have two ethyl groups in identical environments.
















