
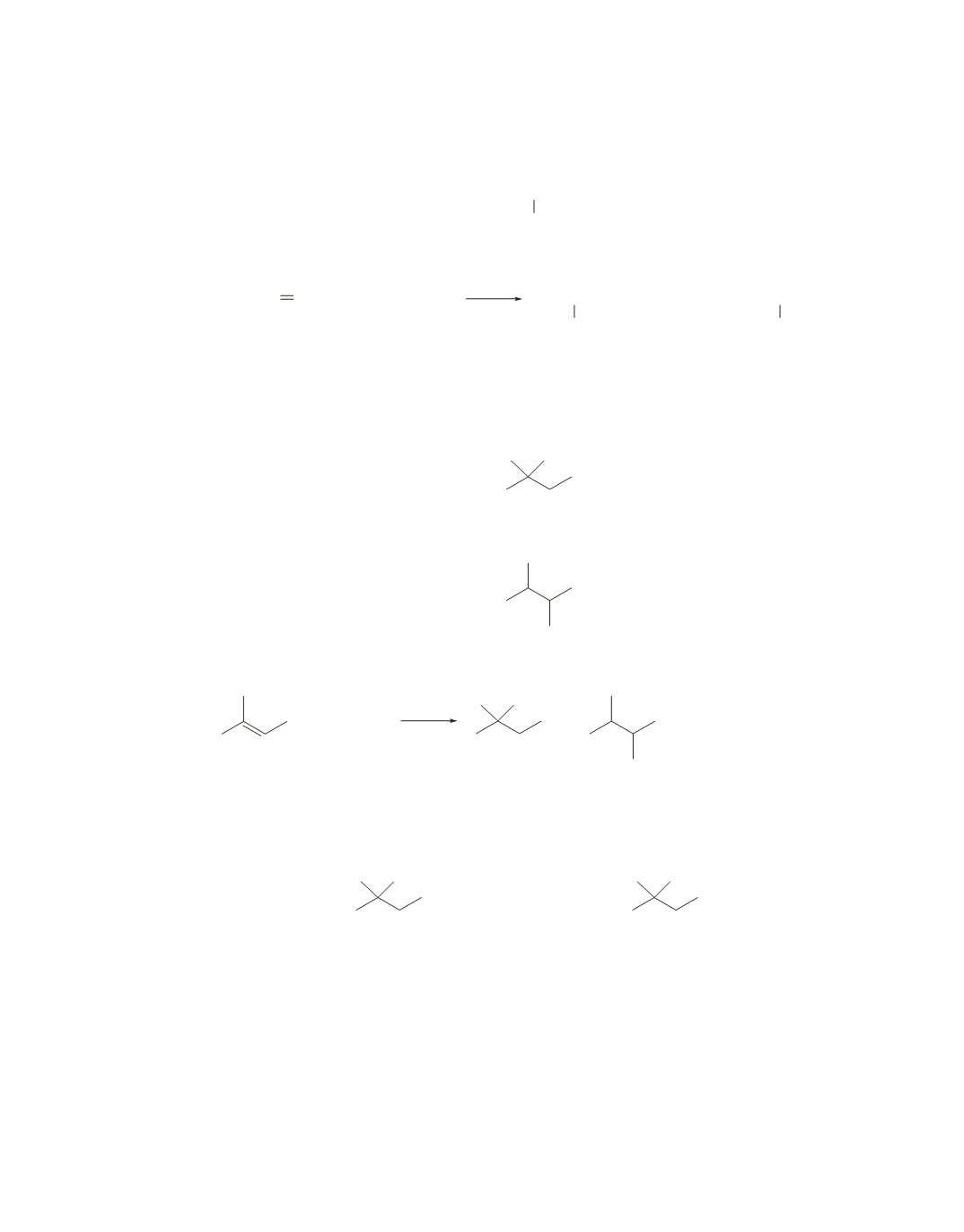
Spectroscopy Problems
69
Copyright © 2017 Pearson Education, Inc.
B.
The 6H triplet at
∼
1.1 ppm indicates two methyl groups in the same environment. There are also two
CH
2
groups in the same environment. Thus, compound
B
is
CH
3
CH
2
CHCH
2
CH
3
Br
The reaction that produces
A
and
B
is
HBr
+
+
CH
3
CH CHCH
2
CH
3
CH
3
CHCH
2
CH
2
CH
3
Br
CH
3
CH
2
CHCH
2
CH
3
Br
C.
The 6H singlet at
∼
2.8 ppm indicates 2 methyl groups that are attached to a carbon that is not attached
to a hydrogen. Because the signal is at a higher frequency than expected for a methyl group, the carbon
must be attached to an electron-withdrawing group (a Br). Integration shows that the compound has a
CH
2
group and another methyl group. Therefore, compound
C
is
Br
D.
The 6H doublet at 0.9 ppm indicates an isopropyl group. The compound has a third methyl group as
well as two carbons that are attached to only one H. Therefore, compound
D
is
Br
The reaction that produces
C
and
D
is
Br
HBr
+
+
Br
58.
The strong absorption band at
∼
3400 cm
-
1
is due to an OH group, which also gives the 1H singlet. The
6H singlet is due to two methyl groups that are attached to a carbon that is not attached to a hydrogen. This
information and the molecular formula give two possible structures for the compound:
OH
Cl
Cl
OH
or
59.
The absence of a molecular ion peak suggests that the compound might be an alcohol. Subtracting 84 from
the molecular ion (102 – 84)
=
18 shows that the peak at
m
>
z
=
84 results from loss of water from the
molecular ion, confirming that the compound is an alcohol.
In order to lose water, the alcohol must have a
g
-hydrogen. The rule of 13 gives a molecular formula of
C
7
H
18
. Because we know that the compound is an alcohol, we must add an O and subtract a C and 4 Hs
from the molecular formula, resulting in a molecular formula of C
6
H
14
O.
















