
64
Spectroscopy Problems
Copyright © 2017 Pearson Education, Inc.
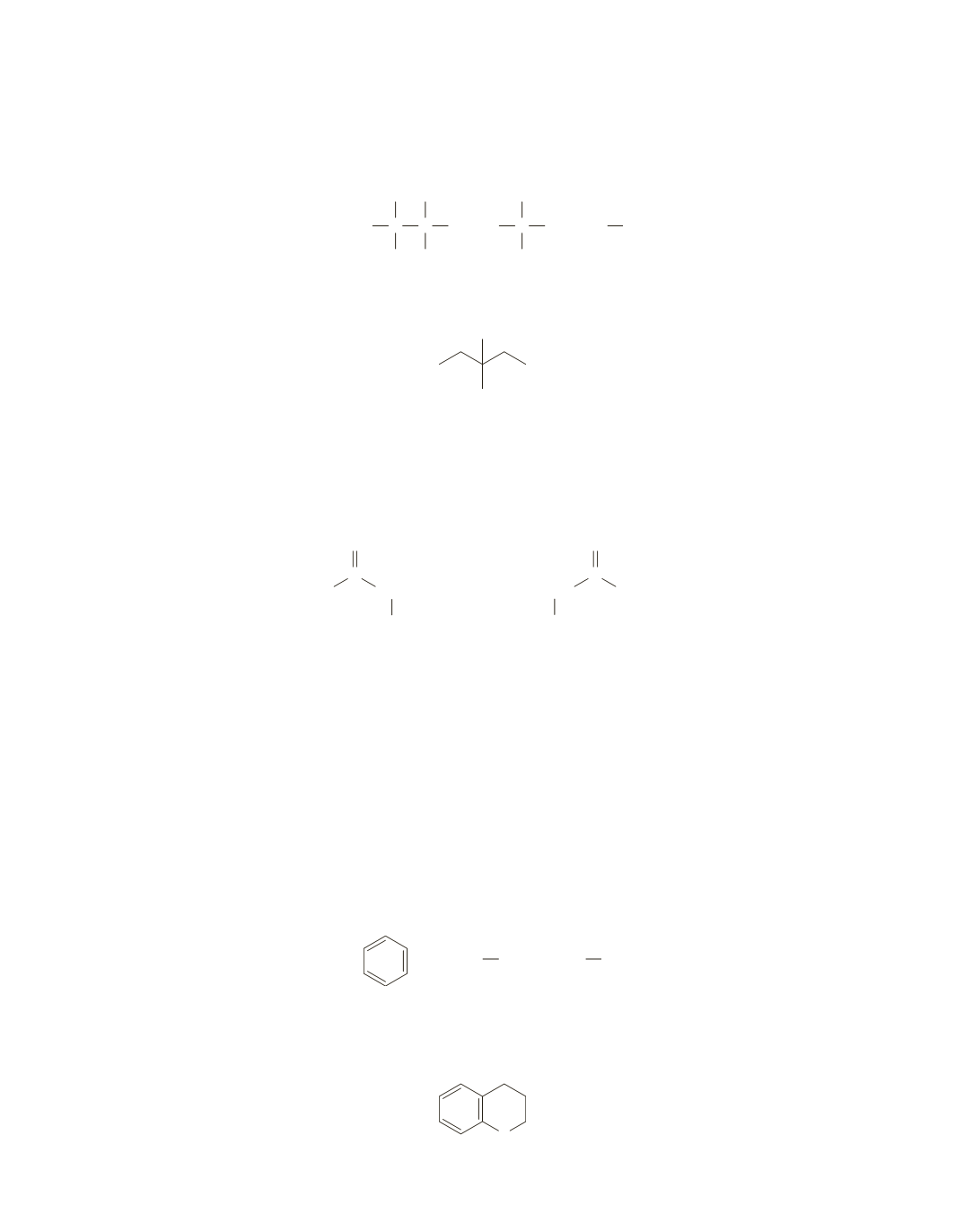
The only other signal in the spectrum is the singlet (1.2 ppm) that integrates to 3 protons. This signal must
be due to a methyl group that is bonded to a carbon that is not attached to any hydrogens.
C
H H
H
H2
C
H
C
H
H
H
HO
Because theNMR spectrumdoes not showany additional signals, the compoundmust be
3-methyl-3-pentanol
.
OH
46.
The doublet (1.1 ppm) that integrates to 6 protons and the septet (~5 ppm) that integrates to 1 proton
suggest an isopropyl group. The triplet (1.7 ppm) that integrates to 3 protons and the quartet (2.2 ppm) that
integrates to 2 protons suggest an ethyl group. When these two groups are subtracted from the molecular
formula, all that remains is
CO
2
.
The splitting patterns tell us that the isopropyl and ethyl groups are
isolated from one another. We can conclude then that the compound is an ester. There are two possibilities:
isopropyl propanoate
ethyl 2-methylpropanoate
or
CH
3
CH
2
OCHCH
3
CH
3
C
O
CH
3
CH OCH
2
CH
2
CH
3
C
O
Because the highest-frequency signal (the septet) is given by the CH of the isopropyl group, we know
that the CH is attached to an oxygen. Therefore, we know that the compound that gives the NMR data is
isopropyl propanoate
.
47.
The multiplet (6.9–7.8 ppm) that integrates to 4 protons indicates a disubstituted benzene ring. Because
the signal is a multiplet, we know that the substituents are on either the 1- and 2-positions or the 1- and
3-positions. If the substituents were on the 1- and 4-positions, either one singlet (if the two substituents are
identical) or two doublets (if the substituents are not identical) would be observed.
Three signals (1.4, 2.5, 3.8 ppm) each integrate to 2 protons. The fact that the signals are two triplets and a
multiplet suggests that the compound has three adjacent methylene groups. (The methylene groups on the
ends will be triplets, and the one in the middle will be a multiplet.)
CH
2
CH
2
CH
2
From the molecular formula, we know that the compound has an oxygen. The triplet at 3.8 ppm indicates
that that particular methylene group is next to the oxygen. The two fragment we have identified account for
the entire molecular formula. Therefore, the compound must have the structure shown here.
O
















