
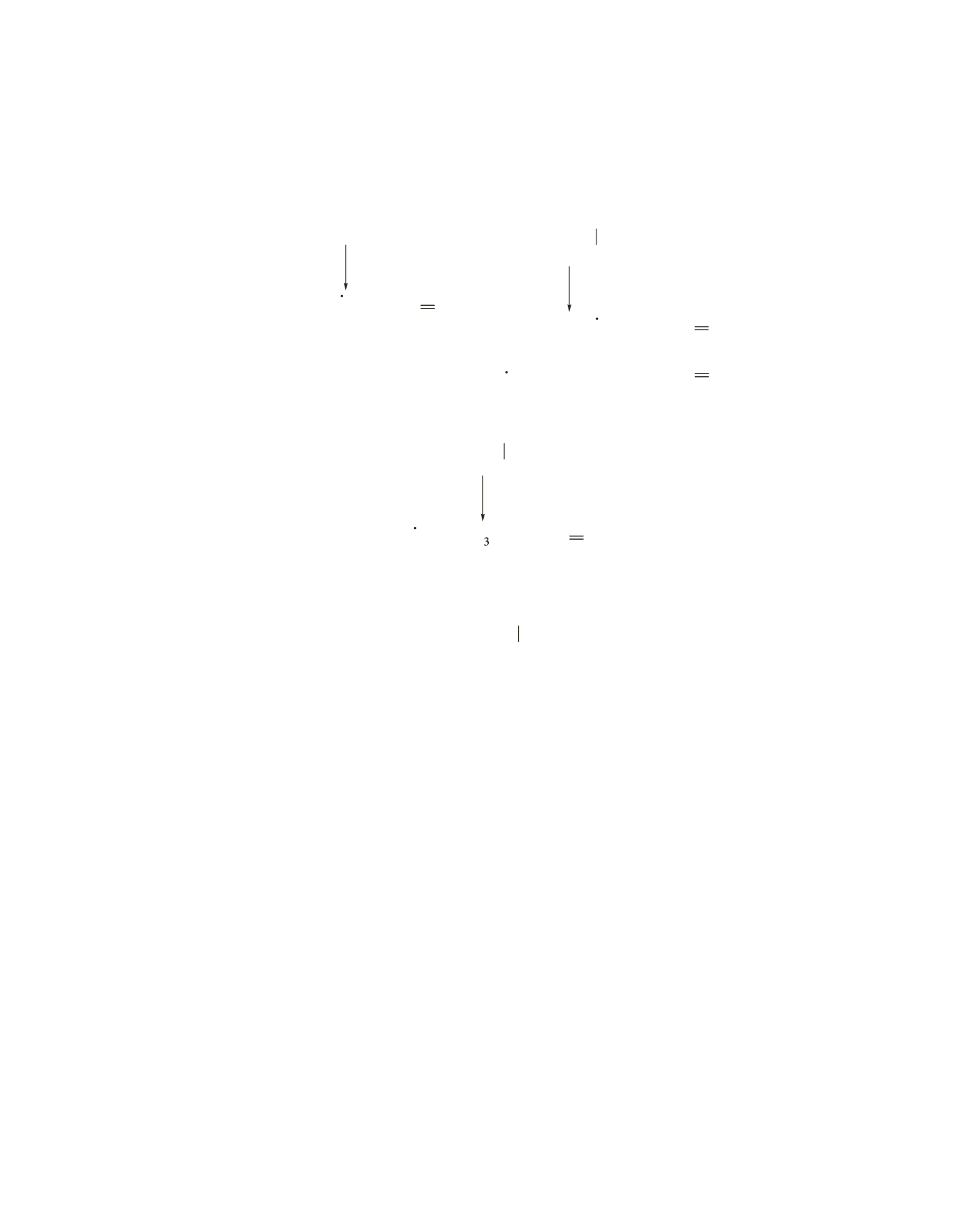
70
Spectroscopy Problems
Copyright © 2017 Pearson Education, Inc.
The molecular ions of three alcohols with molecular formula C
6
H
14
O and a
g
-hydrogen are shown below.
a
-Cleavage of the first alcohol results in a peak at
m
>
z
=
31;
a
-cleavage of the second alcohol results in peaks at
m
>
z
=
45 and 87;
a
-cleverage of the third alcohol results in a peak at
m
>
z
=
73.
CH
3
CH
2
CH
2
CH
3
CH
2
CH
2
CH
m/z
=
45
OH
+
CH CH
3
CH
m/z
=
87
2
CH
2
CH
3
CH
2
CH
2
CH CH
2
CH
3
-cleavage
a
OH
+
OH
+
CH
3
+
CH
3
CH
2
CH
2
CH
m/z
=
31
+
+
CH
2 2
CH
3
CH
2
CH
2
CH CH
2 2
CH
2
OH
-cleavage
a
CH
2
OH
+
+
CH
3
CH
2
CH
2
CHCH
2
CH
2
CH
3
-cleavage
a
CH
3
CH
2
CH
2
CH CH
2
CH
2
CH
m/z
=
73
OH
+
OH
+
+
Therefore, the alcohol that gives the mass spectrum is
CH
3
CH
2
CH
2
CH
OH
CHCH
3
2
60.
A
shows an absorption for a double bond but not for an OH group. Therefore,
A
must be a tertiary
alkyl halide (2-bromo-2-methylpropane) because tertiary alkyl halides undergo only elimination with a
strong base.
B
shows an absorption for an OH group but not for a double bond. Therefore,
B
must be a primary alkyl
halide (1-bromobutane) because primary alkyl halides undergo primarily substitution.
C
shows an absorption for a double bond and for an OH group. Therefore,
C
must be a secondary alkyl
halide (2-bromobutane) because secondary alkyl halides undergo both substitution and elimination.
61.
The strong and broad absorption bands at
∼
3400 cm
-
1
indicate that
A
and
B
are the spectra of alcohols.
The absorptions in Spectrum
A
at a little
6
3000 cm
-
1
indicate hydrogens attached to
sp
2
carbons, and the
absorption at 1600 cm
-
1
indicates a benzene ring. Spectrum
B
has neither of these absorptions. Therefore,
A
is the spectrum of Compound
2
.
B
is the spectrum of Compound
4
.
The strong absorption at
∼
1700 cm
-
1
indicates that
C
,
D
, and
E
are the spectra of compounds with a
carbonyl group. The broad absorption at
∼
3000 cm
-
1
indicates that
E
is the spectrum of a carboxylic acid (Compound
5
).
The absorption at
∼
2700 cm
-
1
indicates that
D
is the spectrum of an aldehyde (Compound
3
). Therefore,
C
is the spectrum of Compound
1
.
















