
Spectroscopy Problems
57
Copyright © 2017 Pearson Education, Inc.
32.
Because all three spectra are given by ethylmethylbenzenes, the low-frequency signals in both the
1
H NMR
and
13
C NMR
spectra can be ignored because they belong to the methyl and ethyl substituents. The key to
determining which spectrum belongs to which ethylmethylbenzene can be found in the aromatic region of
the
1
H NMR
and
13
C NMR
spectra.
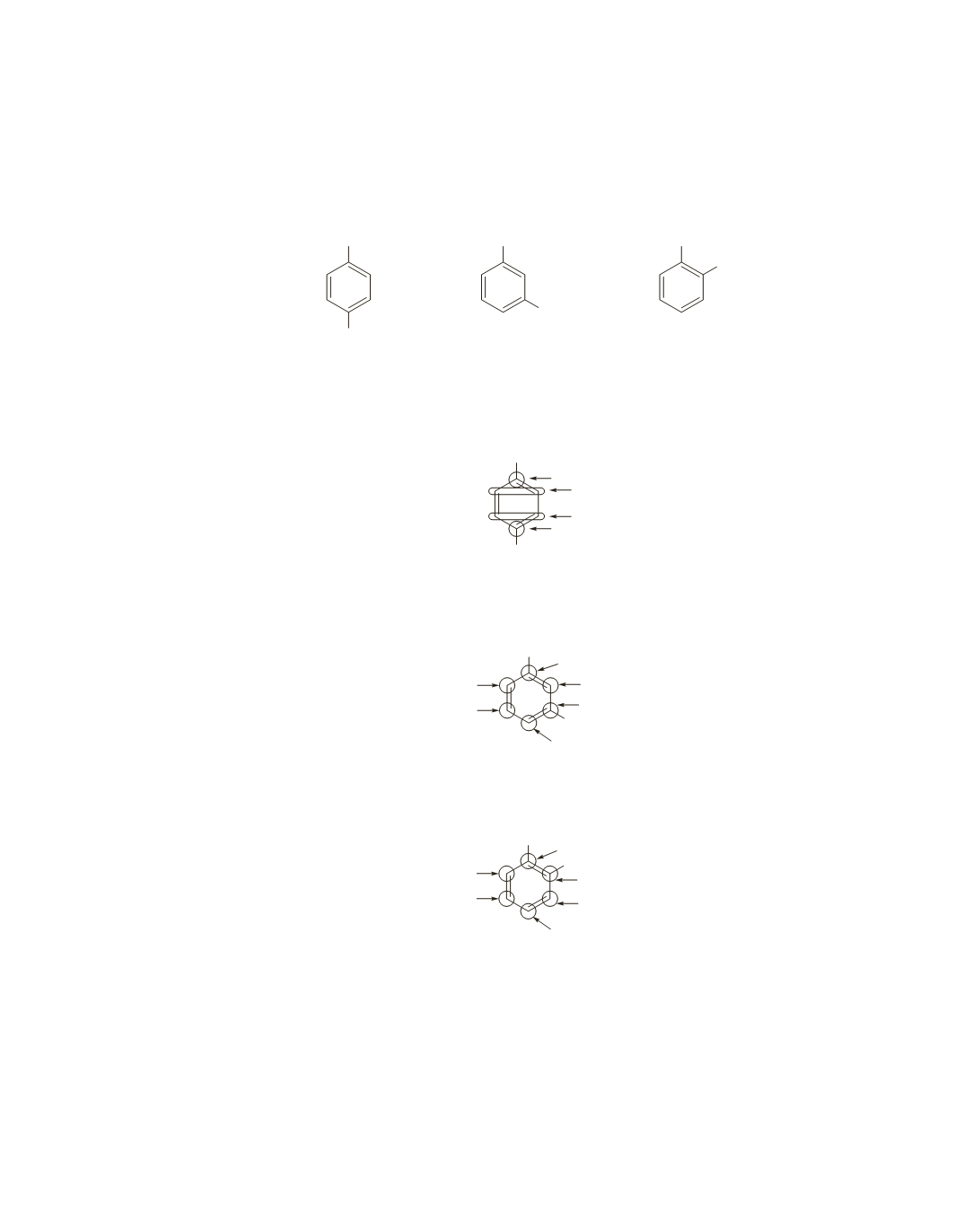
4-ethylmethylbenzene
3-ethylmethylbenzene
2-ethylmethylbenzene
CH
3
CH
2
CH
3
CH
3
CH
2
CH
3
CH
3
CH
2
CH
3
The aromatic region of the
13
C NMR
spectrum of 4-ethylmethylbenzene will show four signals because it
has four different ring carbons.
CH
2
CH
3
#4
#3
#2
#1
CH
3
The aromatic region of the
13
C NMR
spectrum of 3-ethylmethylbenzene will show six signals because it
has six different ring carbons.
#4
#3
#5
#6
#2
#1
CH
2
CH
3
CH
3
The aromatic region of the
13
C NMR
spectrum of 2-ethylmethylbenzene will also show six signals because
it has six different ring carbons.
#4
#3
#5
#6
#2
#1
CH
3
CH
2
CH
3
We now know that spectrum
(b)
is the spectrum of 4-ethylmethylbenzene because its
13
C NMR
spectrum
has four signals and the other two compounds will show six signals.
To distinguish between 2-ethylmethylbenzene and 3-ethylmethylbenzene, we need to look at the splitting
patterns in the aromatic regions of the
1
H NMR
spectra. Analysis of the aromatic region for spectrum
(c)
is
difficult because the signals are superimposed. Analysis of the aromatic region for spectrum
(a)
provides
the needed information. A triplet (7.2 ppm) that integrates to 1 proton is clearly present. This means that
















