
56
Spectroscopy Problems
Copyright © 2017 Pearson Education, Inc.
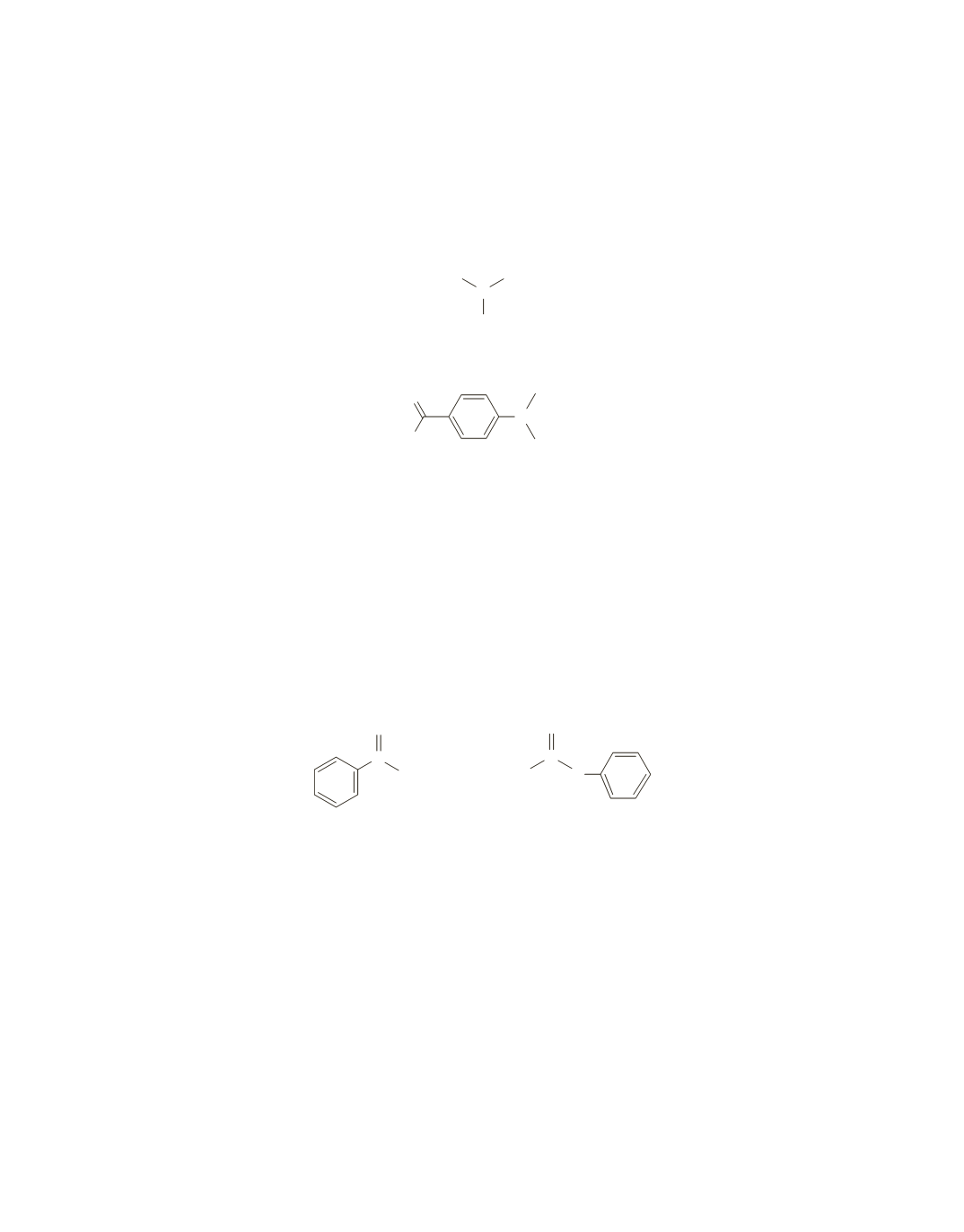
If the aldehyde group and the disubstituted ring are subtracted from the molecular formula, we find that
the second substituent contains 2 carbons, 6 hydrogens, and 1 nitrogen. The remaining NMR signal
( 3.0 ppm)
is a singlet that integrates to 6 hydrogens. These must be due to two methyl groups in the same
environment. The nitrogen must be between the two methyl group; otherwise, they would split each other’s
signals. The nitrogen causes the signal for the methyl groups to appear at a higher frequency than where
methyl groups normally appear.
N
CH
3
H
3
C
Thus, the compound is
4-(dimethylamino)benzaldehyde
.
H
O
CH
3
CH
3
N
31.
The three signals between 7.4 and 8.1 ppm that together integrate to 5 protons indicate a monosubstituted
benzene ring. Subtracting the monosubstituted ring
(C
6
H
5
)
from the molecular formula leaves
C
3
H
5
O
2
to
be accounted for.
The two oxygens in the molecular formula tells us that the compound is an ester because a broad singlet
between 10 and 12 ppm that would indicate a carboxylic acid is not present. The remainder of the molecule
contains two carbons and five hydrogens.
The two remaining signals, a triplet (1.4 ppm) that integrates to 3 protons and a quartet (4.4 ppm) that
integrates to 2 protons, are characteristic of an ethyl group. The three known segments can now be joined
in one of two ways:
ethyl benzoate
OCH
2
CH
3
C
O
phenyl propanoate
O
CH
3
CH
2
C
O
The choice between the two compounds can be made by looking at the chemical shift of the methylene
protons. In the ethyl ester, the signal will be highly deshielded by the adjacent oxygen. In the phenyl ester,
the signal will be at
2.1 ppm
because the methylene protons are next to the carbonyl group. Because the
chemical shift of the methylene protons is 4.4 ppm, we know that the compound is
ethyl benzoate
.
















