
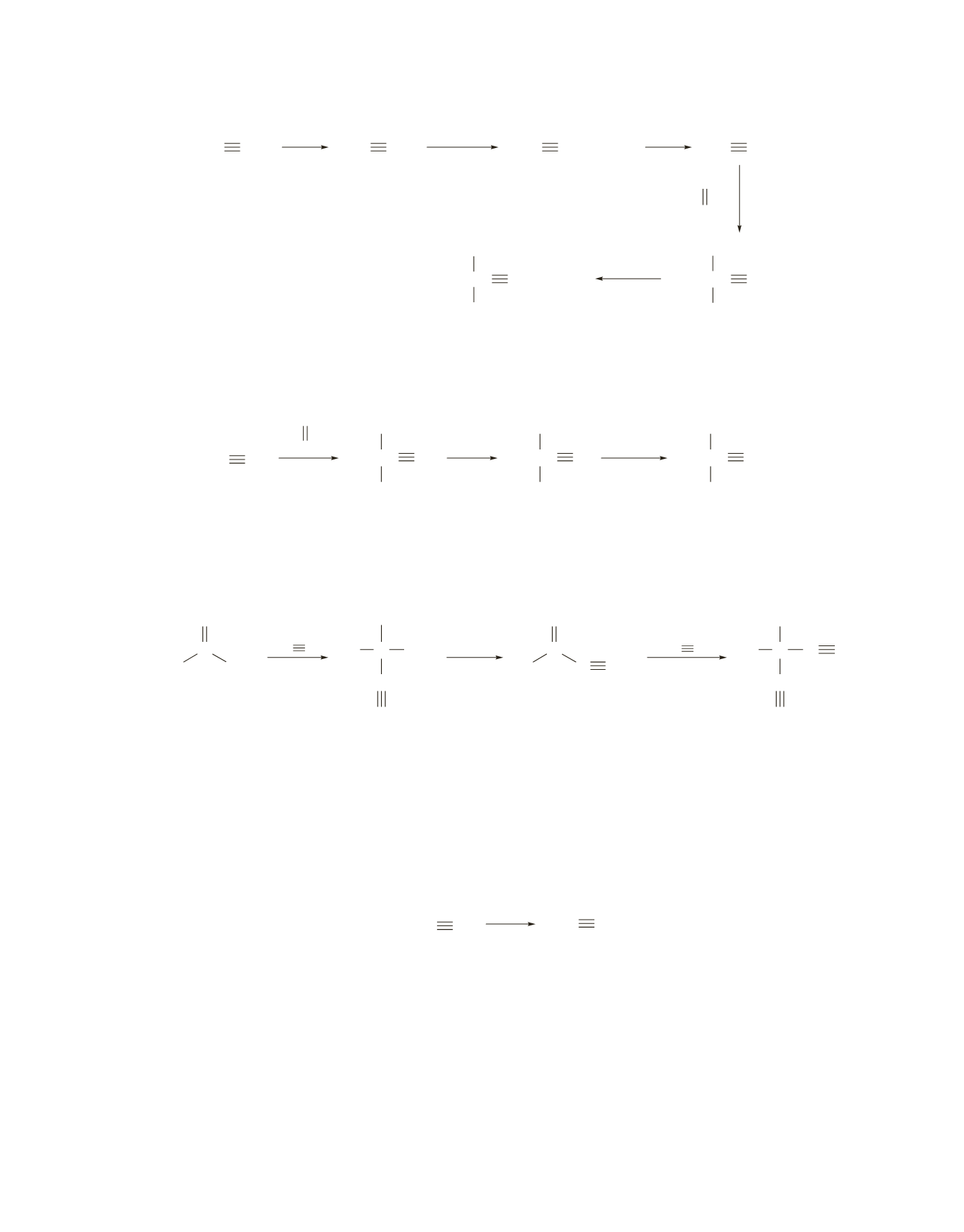
Chapter 16 521
Copyright © 2017 Pearson Education, Inc.
c.
HC CH
HC C
−
CH
3
CH
2
Br
HC CCH
2
CH
3
C CCH
2
CH
3
−
CH
3
CCH
3
O
CH
3
CC CCH
2
CH
3
CH
3
O
−
CH
3
CC CCH
2
CH
3
CH
3
OH
NHNa
2
NHNa
2
pyridinium
chloride
Notice that ethyne must be alkylated before it reacts as a nucleophile with the carbonyl compound. If
ethyne is alkylated after nucleophilic addition to the ketone, alkylation can occur on both carbon and
oxygen.
CH
3
CCH
3
O
NaNH
2
CH
3
CH
2
Br
−
C CH
CH
3
CC CH
CH
3
O
−
CH
3
CC C
−
CH
3
O
−
CH
3
CC
CCH
2
CH
3
CH
3
OCH
2
CH
3
14.
Similar to the way a Grignard reagent reacts with an ester, an acetylide ion first reacts with the ester in a
nucleophilic acyl substitution reaction to form a ketone; this is followed by a nucleophilic addition reaction
to form a tertiary alcohol.
R OR
C
O
RC C
−
R OR C
O
−
C
C
R
R C
C
O
CR
R CC
OH
C
C
R
CR
+
RO
−
RC C
−
a ketone
a tertiary alcohol
1.
2. pyridinium
chloride
Notice that the alkoxide ion is protonated by pyridinium chloride rather than by HCl or
H
3
O
+
because
these acids would add to the triple bonds.
15.
The reaction is carried out with excess cyanide ion in order to have some unprotonated cyanide ion to act as
a nucleophile. HCl is a strong acid, so it dissociates completely and protonates the cyanide ion. Therefore,
there is no HCl in the reaction mixture, so HCN is the only acid available to protonate the alkoxide ion.
HCl
HC N
−
C N
+
+
Cl
−
16.
No, an acid must be present in the reaction mixture to protonate the oxygen of the cyanohydrin. Otherwise,
the cyano group will be eliminated and the reactants will reform.
17.
Strong acids like HCl and
H
2
SO
4
have very weak conjugate bases
1
Cl
-
and
HSO
4
-
2
, which are excel-
lent leaving groups. When these bases add to the carbonyl group, they are readily eliminated, reforming
the starting materials. Cyanide ion is a strong enough base not to be eliminated unless the oxygen in the
product is negatively charged.
18.
Solved in the text.
















