
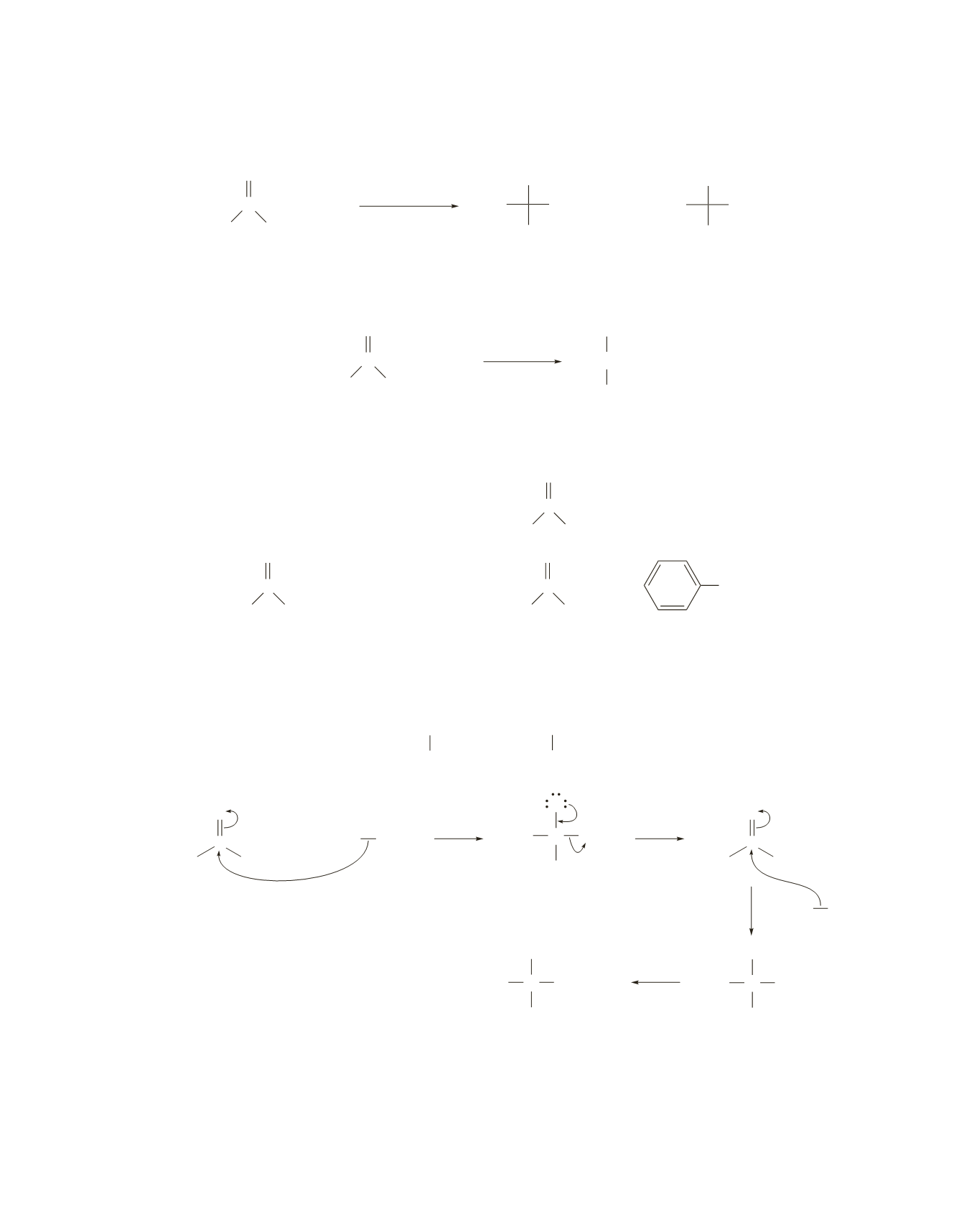
Chapter 16 519
Copyright © 2017 Pearson Education, Inc.
7.
a.
Two stereoisomers are obtained, because the reaction creates an asymmetric center in the product.
CH
3
OH
CH
2
CH
2
CH
3
CH
2
CH
3
CH
3
CH
2
CH
3
CH
2
CH
2
CH
3
OH
1. CH
3
CH
2
MgBr
2. H
3
O
+
(
S
)-3-methyl-3-hexanol
(
R
)-3-methyl-3-hexanol
CH
3
CH
2
CH
2
CH
3
C
O
b.
Only one compound is obtained, because the product does not have an asymmetric center.
1. CH
3
MgBr
CH
3
CCH
2
CH
2
CH
3
OH
CH
3
CH
3
CH
2
CH
2
CH
3
C
O
2. H
3
O
+
8.
a.
Solved in the text.
b.
B
CH
3
OR
2 CH
3
MgBr
+
A
Solved in the text
.
D
2 CH
3
CH
2
MgBr
+
F
2
+
MgBr
C
O
CH
3
OR
C
O
CH
3
OR
C
O
9.
If a secondary alcohol is formed from the reaction of a formate ester with excess Grignard reagent, the
two alkyl substituents of the alcohol will be identical because they both come from the Grignard reagent.
Therefore, only the following two alcohols (
B
and
D
) can be prepared that way.
CH
3
CHCH
3
OH
CH
3
CH
2
CHCH
2
CH
3
OH
10.
+
O
Cl
CH
3
C
O
CH
2
CH
3
CH
3
C
CH
3
CH
2
MgBr
CH
3
C Cl
CH
2
CH
3
O
−
CH
3
C CH
2
CH
3
CH
2
CH
3
OH
CH
3
C CH
2
CH
3
CH
3
CH
2
MgBr
CH
2
CH
3
O
−
H
3
O
+
+
MgBr
+
MgBr
















