
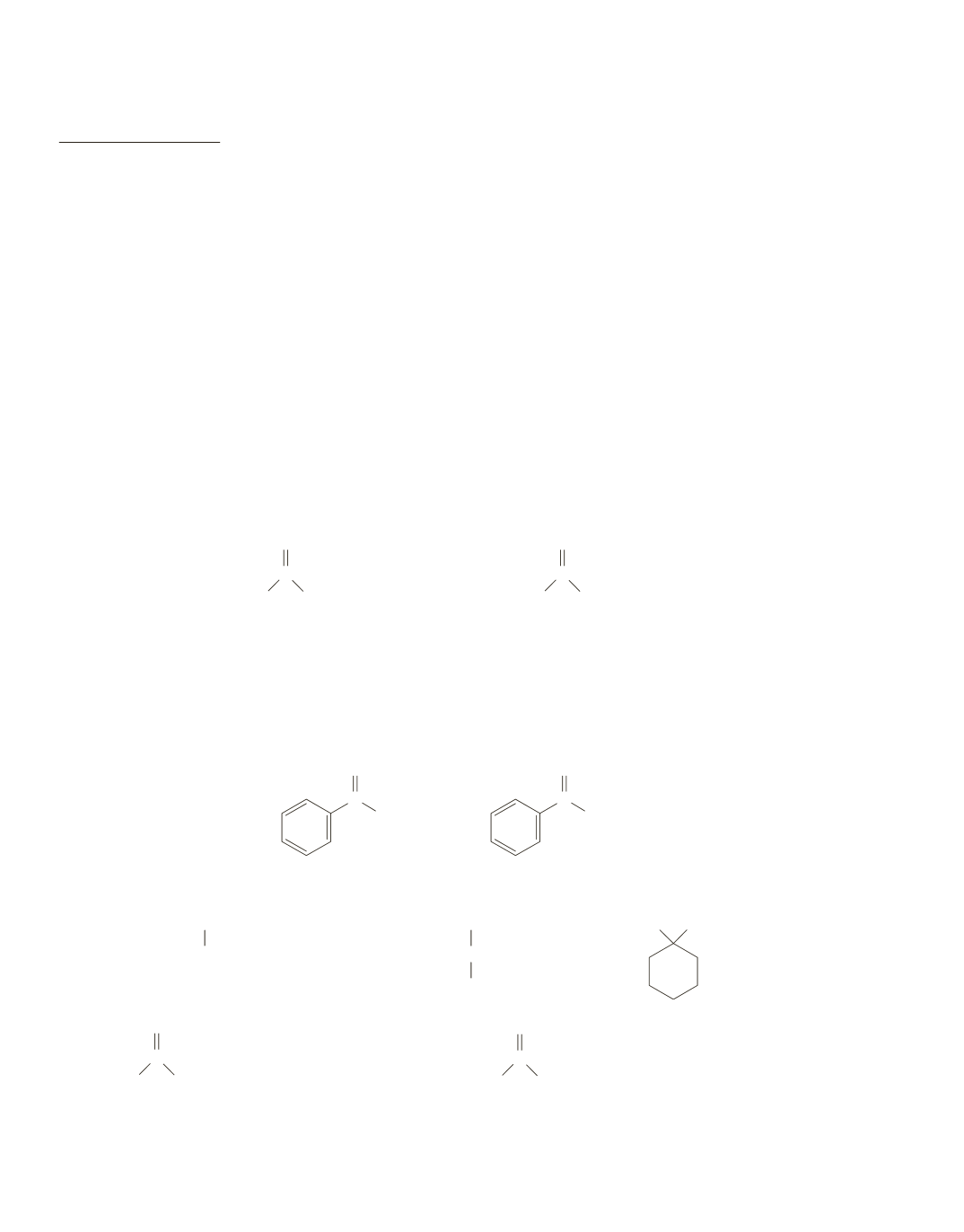
518 Chapter 16
Copyright © 2017 Pearson Education, Inc.
Solutions to Problems
1.
a.
3-methylpentanal,
b
-methylvaleraldehyde
b.
4-heptanone, dipropyl ketone
c.
2-methyl-4-heptanone, isobutyl propyl ketone
d.
4-phenylbutanal,
g
-phenylbutyraldehyde
e.
4-ethylhexanal,
g
-ethylcaproaldehyde
f.
1-hepten-3-one, butyl vinyl ketone
2.
If the carbonyl group were anywhere else in these compounds, they would not be ketones (they would be
aldehydes) and, therefore, would not have the “one” suffix.
3.
a.
6-hydroxy-3-heptanone
b.
2-oxocyclohexylmethanenitrile
c.
4-formylhexanamide
4.
a.
2-Heptanone is more reactive because it has less steric hindrance. There is little difference in the
amount of steric hindrance provided by the propyl and the pentyl group at the carbonyl carbon (the site
of nucleophilic addition) because they differ at a point somewhat removed from the site of nucleophilic
addition. However, there is a significant difference in size between a methyl group and a propyl group
at the site of nucleophilic addition.
CH
3
CH
2
CH
2
CH
2
CH
2
CH
3
CH
3
CH
2
CH
2
CH
2
CH
2
CH
3
2-heptanone
4-heptanone
C
O
C
O
b.
Chloromethyl phenyl ketone is more reactive because chlorine is more strongly electron withdrawing
than bromine since chlorine is more electronegative. Withdrawing electrons inductively away from the
carbonyl group makes the carbonyl carbon more electrophilic and, therefore, more reactive toward a
nucleophile.
C
CH
2
Br
O
bromomethyl phenyl ketone
chloromethyl phenyl ketone
C
CH
2
Cl
O
5.
a.
CH
3
CH
2
CHCH
3
OH
b.
CH
3
CH
2
CH
2
CCH
3
OH
CH
3
c.
HO CH
3
6.
CH
3
CH
2
CH
3
+
CH
3
CH
2
CH
2
MgBr
CH
3
CH
2
CH
2
CH
2
CH
3
CH
3
MgBr
+
or
C
O
C
O
















