
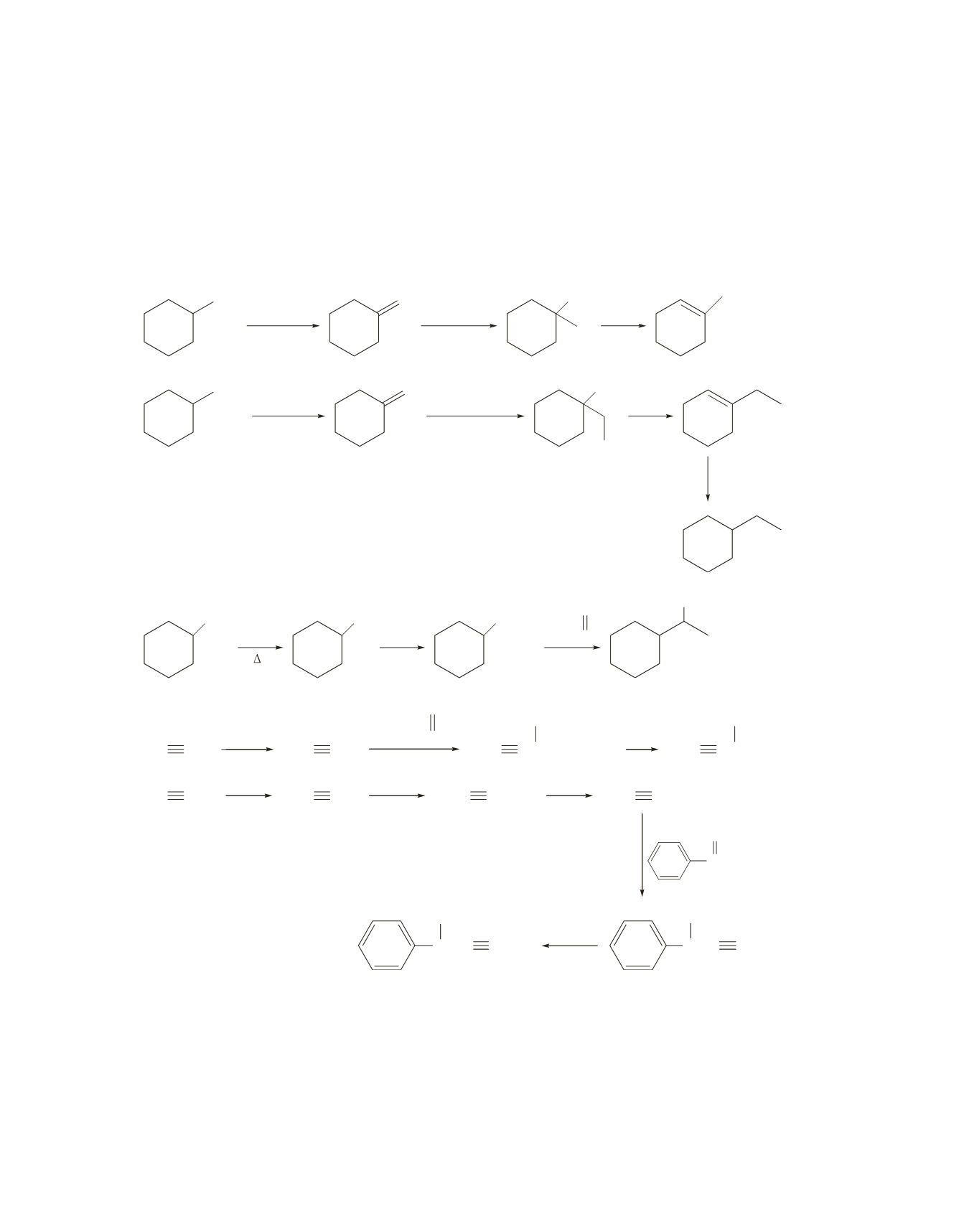
520 Chapter 16
Copyright © 2017 Pearson Education, Inc.
11.
A
and
C
will not undergo nucleophilic addition with a Grignard reagent.
A
has an H bonded to a nitrogen,
and
C
has an H bonded to an oxygen; these acidic hydrogens will react rapidly with the Grignard reagent,
converting it to an alkane before the Grignard reagent has a chance to react as a nucleophile.
B
will undergo a nucleophilic addition–elimination reaction with the Grignard reagent to form a ketone.
This will be followed by a nucleophilic addition reaction of the ketone with another equivalent of the
Grignard reagent. The product will be a tertiary alcohol.
12.
a.
NaOCl
CH
3
COOH
0 °C
OH
O
1. CH
3
MgBr
2. H
3
O
+
H
2
SO
4
∆
OH
b.
NaOCl
CH
3
COOH
0 °C
OH
O
1. CH
3
CH
2
MgBr
2. H
3
O
+
H
2
SO
4
H
2
Pd/C
∆
OH
c.
H
3
O
+
2.
HBr
Mg
Et
2
O
MgBr
Br
OH
OH
CH
3
CH 1.
O
13.
a.
HC CH
HC C
−
HC CCHCH
2
CH
3
O
−
HC CCHCH
2
CH
3
OH
HCl
NHNa
2
CH
3
CH
2
CH
O
b.
HC CH
HC C
−
HC CCH
3
C CCH
3
−
CH
3
Br
CH
O
CHC CCH
3
CHC CCH
3
O
−
OH
NHNa
2
NHNa
2
pyridinium
chloride
Pyridinium chloride is used to protonate the alkoxide ion in the last step because it will not add to the
triple bond like HCl will.
















