
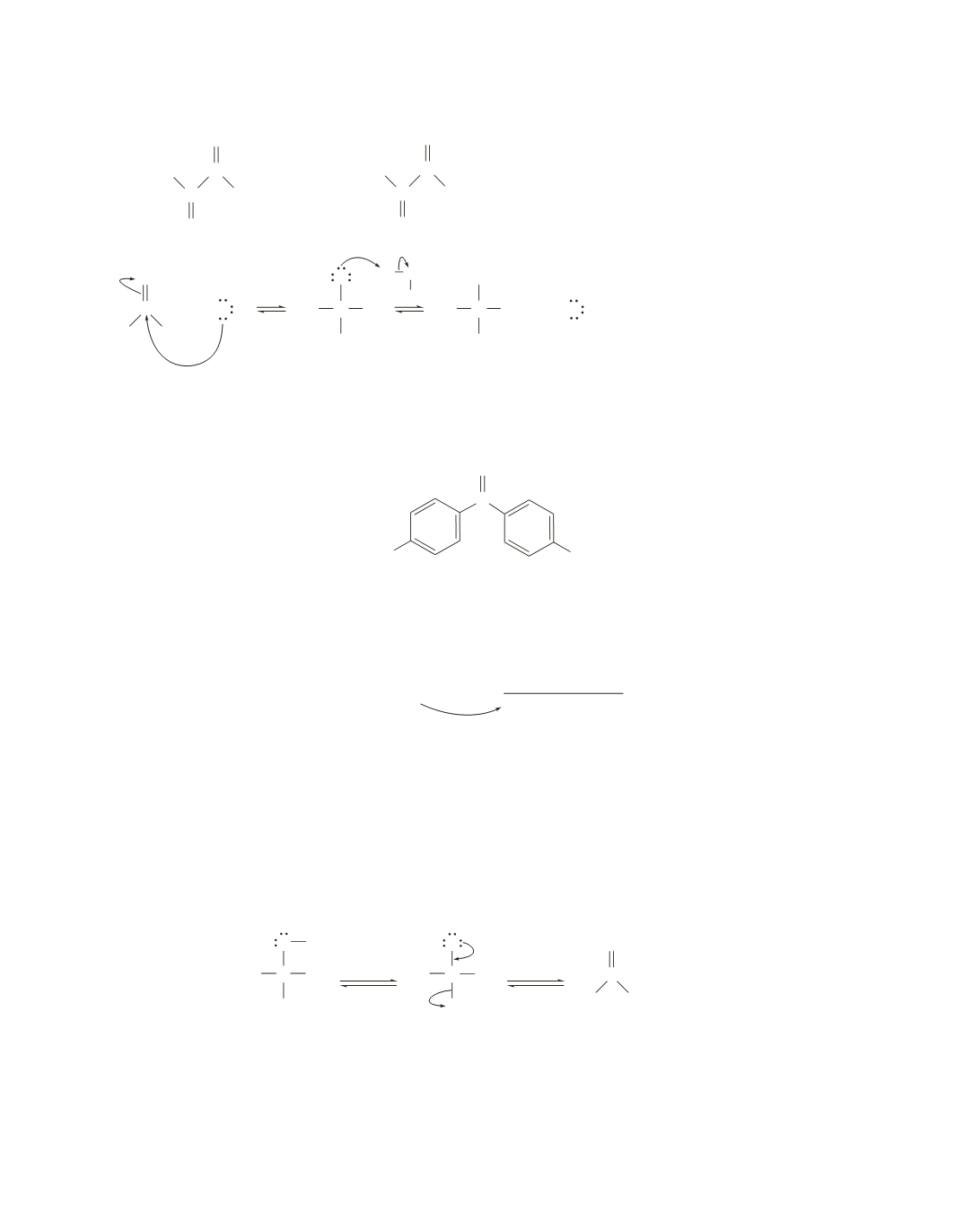
526 Chapter 16
Copyright © 2017 Pearson Education, Inc.
35.
a.
CH
3
O
C
C
O
−
O
b.
O
C
C
O
−
(CH
3
)
2
CH
O
36.
H R
+
HO
−
R C H
O
OH
−
H O
H
R C H
OH
OH
+
HO
−
C
O
37.
Because an electron-withdrawing substituent decreases the stability of a ketone and increases the stability
of a hydrate, the compound with the electron-withdrawing nitro substituents has the largest equilibrium
constant for addition of water.
C
O
NO
2
O
2
N
38.
The electron-withdrawing chlorines decrease the stability of the aldehyde (and, therefore, decrease
its concentration), increasing the equilibrium constant for hydrate formation compared to, for instance,
acetaldehyde.
K
eq
2
hydrate
aldehyde H O
=
[
]
[
] [
]
the concentration
is decreased
39.
a.
hemiacetals: 1, 7, 8
b.
acetals: 2, 3, 5
c.
hydrates: 4, 6
40.
a.
Hemiacetals are unstable in basic solution because the base can remove a proton from an OH group,
thereby providing an oxyanion that can expel the OR group (since the charge that develops on the
oxygen in the transition state is negative) and form a stable aldehyde.
HO
−
R C H
O
OR
−
R C H
O H
OR
+
RO
−
R H
O
C
















