
Chapter 12 419
Copyright © 2017 Pearson Education, Inc.
27.
a.
CH
3
CCH
3
CH
3
CH
3
dimethylpropane
b.
CH
3
CH
2
CHCH
2
CH
2
CH
3
CH
3
3-methylhexane
28.
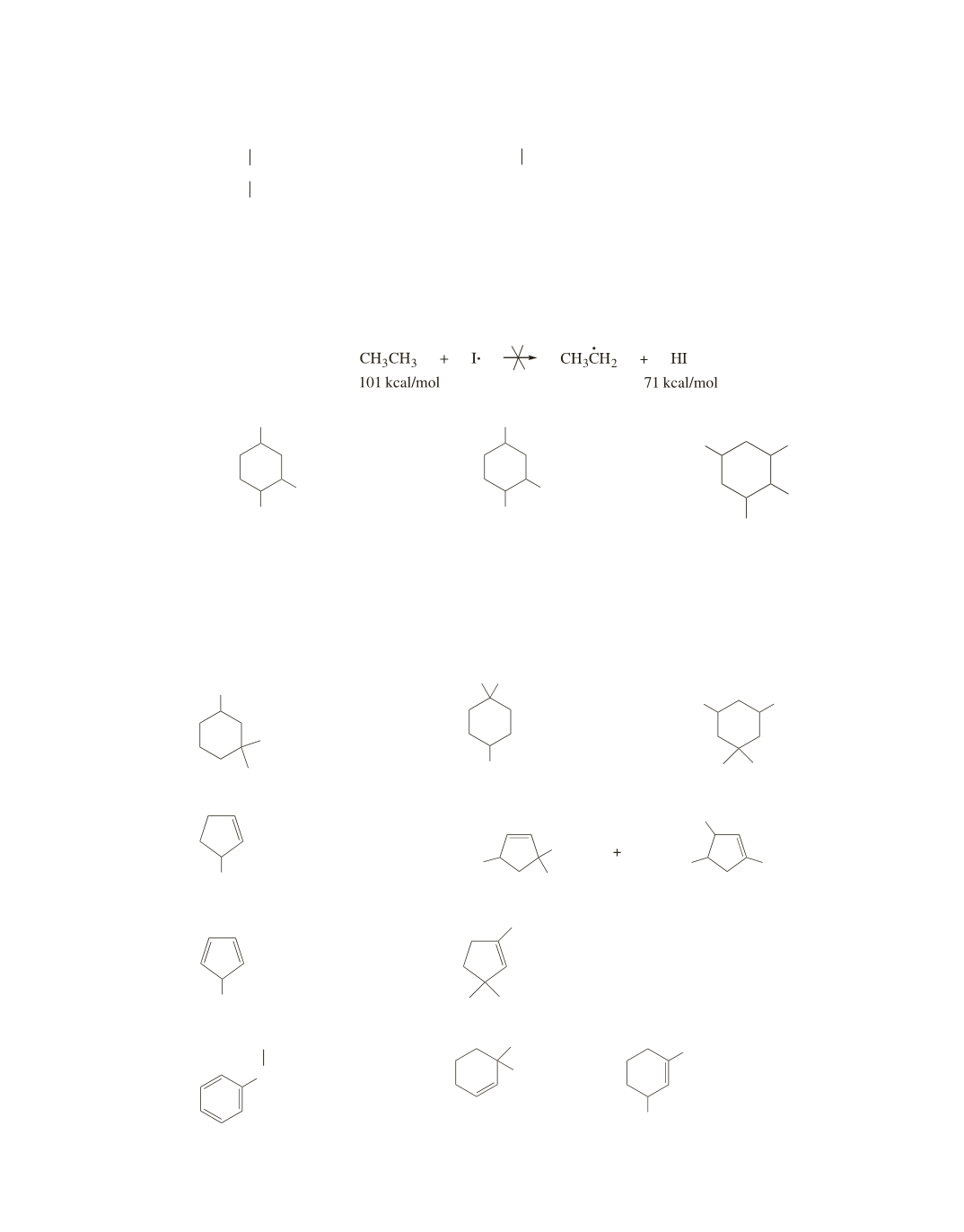
Removing a hydrogen atom from ethane by an iodine radical is a highly endothermic reaction
(
∆
H
°
=
101
-
71
=
30 kcal
>
mol; see Table 5.1 on page 206 of the text), so the iodine radicals will
reform I
2
rather than remove a hydrogen atom.
29.
a.
Four secondary hydrogens
can be removed to form
this product.
CH
3
Cl
CH
3
b.
c.
CH
3
Cl
H
3
C
CH
3
CH
3
CH
3
Cl
Eight secondary
hydrogens can be
removed to form
this product.
Six secondary
hydrogens can be
removed to form
this product.
c.
c.
CH
3
Cl
H
3
C
CH
3
CH
3
CH
3
Cl
Eight secondary
hydrogens can be
removed to form
this product.
Six secondary
hydrogens can be
removed to form
this product.
30.
Because a bromine radical is more selective than a chlorine radical, the bromine radical will remove a
tertiary hydrogen to form a tertiary radical.
a.
CH
3
Br
CH
3
b.
CH
3
CH
3
Br
c.
H
3
C
CH
3
Br CH
3
31.
a.
Br
d.
CH
3
CH
3
Br
CH
3
CH
3
Br
b.
Br
e.
CH
3
CH
3
Br
c.
CHBr
CH
3
f.
CH
3
Br
CH
3
Br
+
















