
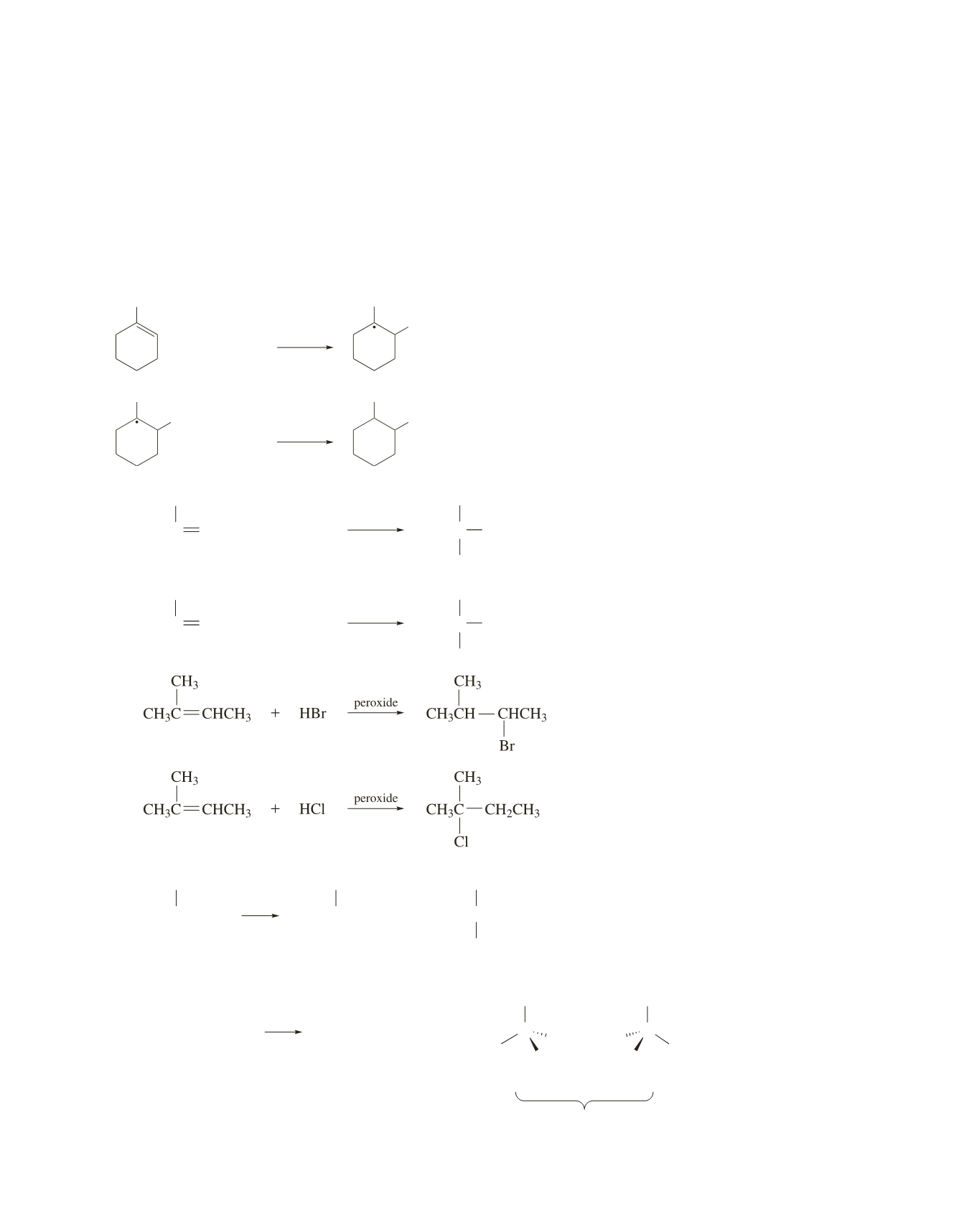
414 Chapter 12
Copyright © 2017 Pearson Education, Inc.
12.
To start peroxide formation, the chain-initiating radical removes a hydrogen atom from an
a
-carbon of the
ether.
a. D
is most apt to form a peroxide, because removal of a hydrogen from an
a
-carbon forms a secondary
radical.
b. B
is least apt to form a peroxide, because it does not have any hydrogens bonded to its
a
-carbons.
13.
CH
3
•
Br
+
CH
3
Br
CH
3
HBr
+
CH
3
Br
Br
Br
+
•
14.
a.
CH
3
C CHCH
3
CH
3
+
HBr
CH
3
C CH
2
CH
3
CH
3
Br
CH
3
C CHCH
3
CH
3
+
HCl
CH
3
C CH
2
CH
3
CH
3
Cl
b.
CH
3
C CHCH
3
CH
3
+
HBr
CH
3
C CH
2
CH
3
CH
3
Br
CH
3
C CHCH
3
CH
3
+
HCl
CH
3
C CH
2
CH
3
CH
3
Cl
c.
d.
15.
a.
CH
3
CHCH
3
+
Cl
2
hv
CH
3
CH
3
CHCH
2
Cl
achiral
CH
3
CH
3
CCH
3
achiral
CH
3
Cl
b.
CH
3
CH
2
CH
2
CH
3
CH
3
CH
2
CH
2
CH
2
Cl
achiral
+
Cl
2
hv
C
CH
2
CH
3
CH
3
Cl
H
C
CH
2
CH
3
CH
3
Cl
H
+
chiral
chiral
enantiomers
















