
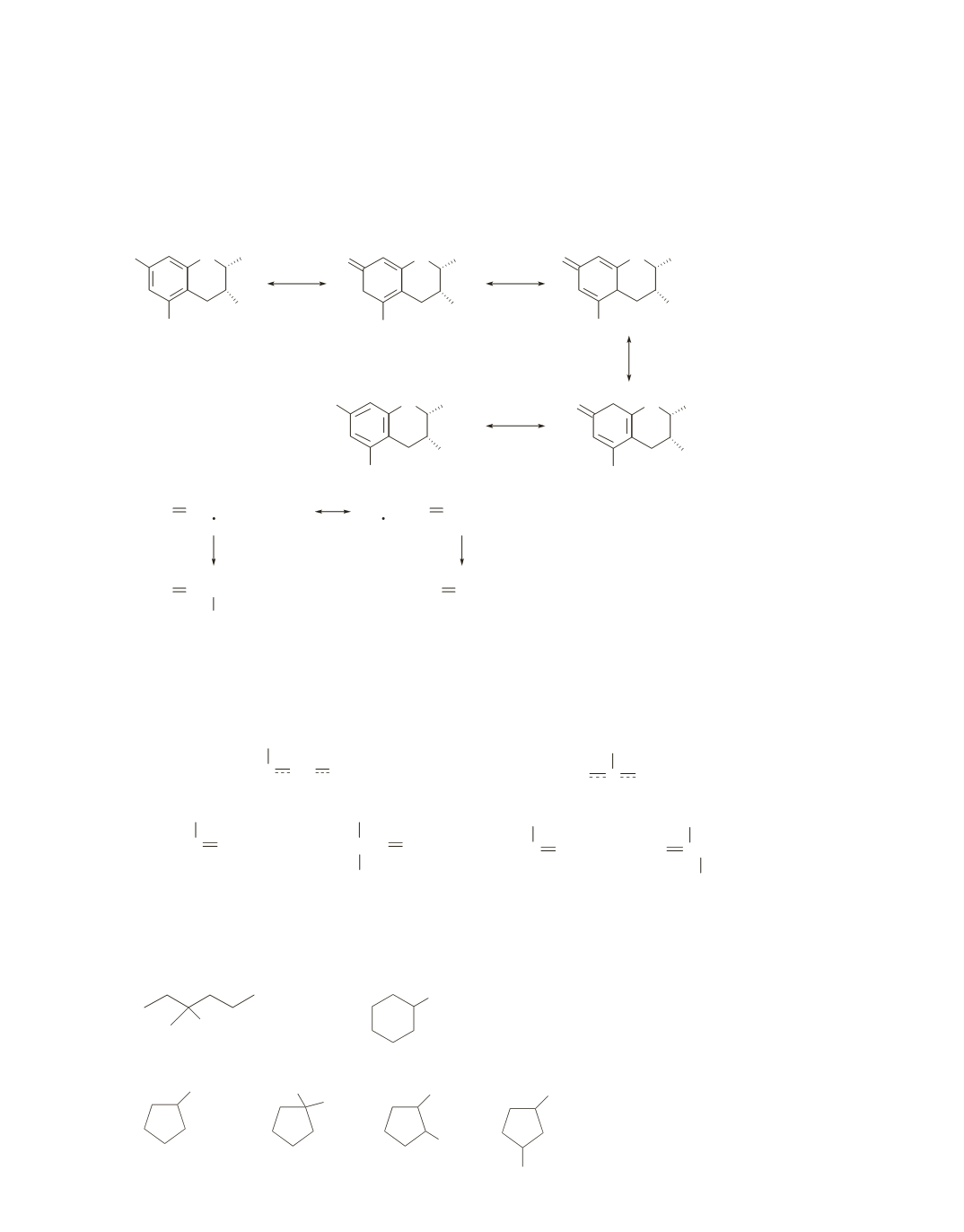
418 Chapter 12
Copyright © 2017 Pearson Education, Inc.
25.
Antioxidants are radical inhibitors. That is, they react with radicals and, thereby, prevent radical chain
reactions. There are several OH groups in a catechin that upon losing a hydrogen atom as a result of react-
ing with a reactive radical, form a radical that is stabilized by electron delocalization. Because this highly
stabilized radical is sufficiently unreactive, it cannot damage cells by reacting with them. An example of
one of the stabilized radicals is shown below.
OH
O
OH
O
OH
O
OH
O
OH
O
.
.
.
O
OH
O
OH
O
OH
O
OH
O
OH
.
.
26.
a.
CH
2
CHCHCH
2
CH
3
CH
2
CH CHCH
2
CH
3
Br
2
Br
Br
2
CH
2
CHCHCH
2
CH
3
BrCH
2
CH CHCH
2
CH
3
+
b.
In the radical intermediate that leads to the major products, the unpaired electron is shared by a
primary carbon and a tertiary carbocation. In the radical intermediate that leads to the minor products,
the unpaired electron is shared by a primary and a secondary carbocation. It, therefore, is not as stable
as the intermediate that leads to the major products.
CH
3
C CH CH
2
CH
3
•
•
CH
3
C CHCH
2
Br
CH
3
+
CH
3
CCH CH
2
Br
CH
3
major products
CH
2
C CHCH
3
•
•
BrCH
2
C CHCH
3
CH
3
+
CH
2
CCHCH
3
minor products
CH
3
CH
3
Br
In bromination, selectivity is more important than probability. Therefore, even though twice as many
hydrogens are available for removal by a bromine radical that leads to the minor products, they will
still be minor products because the easier-to-remove hydrogens lead to the major products.
c.
Br
major product
d.
Cl
e.
no reaction
(There is no light or heat,
so radicals cannot be formed.)
f.
CH
2
Cl
Cl
CH
3
CH
3
Cl
CH
3
Cl
+
+
+
















