
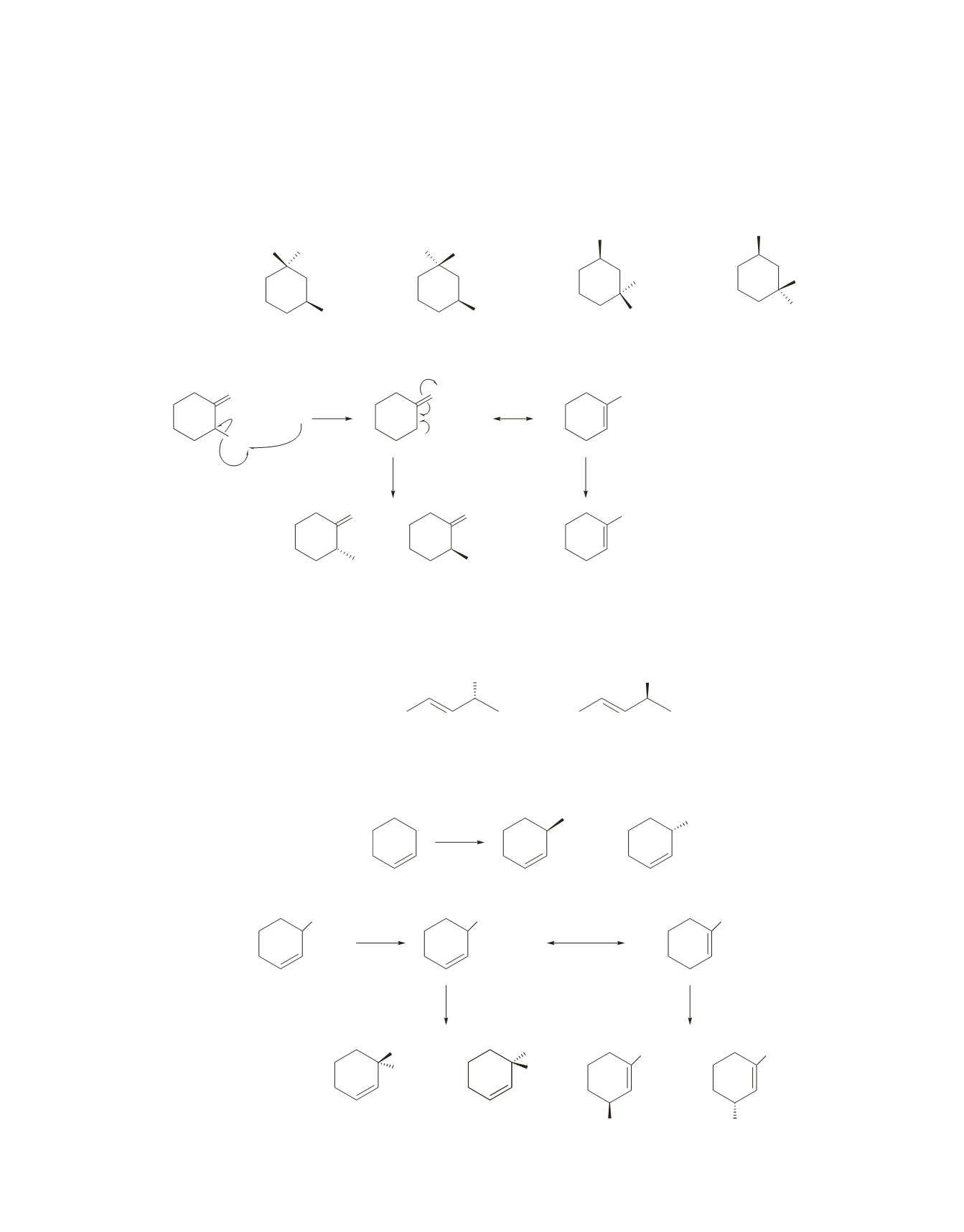
Chapter 12 415
Copyright © 2017 Pearson Education, Inc.
16.
The major monobromination product results from bromination at a tertiary carbon. Bromination can occur
at either of the two tertiary carbons. Because the radical intermediate is
sp
2
hybridized and, therefore, pla-
nar, the incoming bromine can add to a tertiary radical from either the top or bottom of the plane. Notice
that the product (1-bromo-1,3-dimethylcyclohexane) has two asymmetric centers; therefore, four stereo-
isomers are formed:
R,R, R,S, S,S,
and
S,R
.
Br
Br
Br
Br
17.
Solved in the text.
18.
CH
2
CH
2
H
+
+
Br
•
•
CH
2
CH
2
•
Br
2
Br
2
CH
2
Br
Br
CH
2
Br
+
HBr
19.
The product of the reaction in Problem 17 has one asymmetric center. Therefore, two allylic substituted
bromoalkenes are obtained, one with the
R
configuration at the asymmetric center and one with the
S
configuration.
Br
(
R
)-4-bromo-2-pentene
Br
(
S
)-4-bromo-2-pentene
20.
a.
Two stereoisomers are formed because the reaction forms a compound with an asymmetric center.
Br
Br
NBS,
peroxide
+
b.
The reaction forms two constitutional isomers, and each constitutional isomer has two stereoisomers.
CH
3
CH
3
CH
3
CH
3
CH
3
HBr
Br
2
Br
Br
2
NBS,
peroxide
+
+
Br
Br
CH
3
Br
CH
3
+
•
•
















