
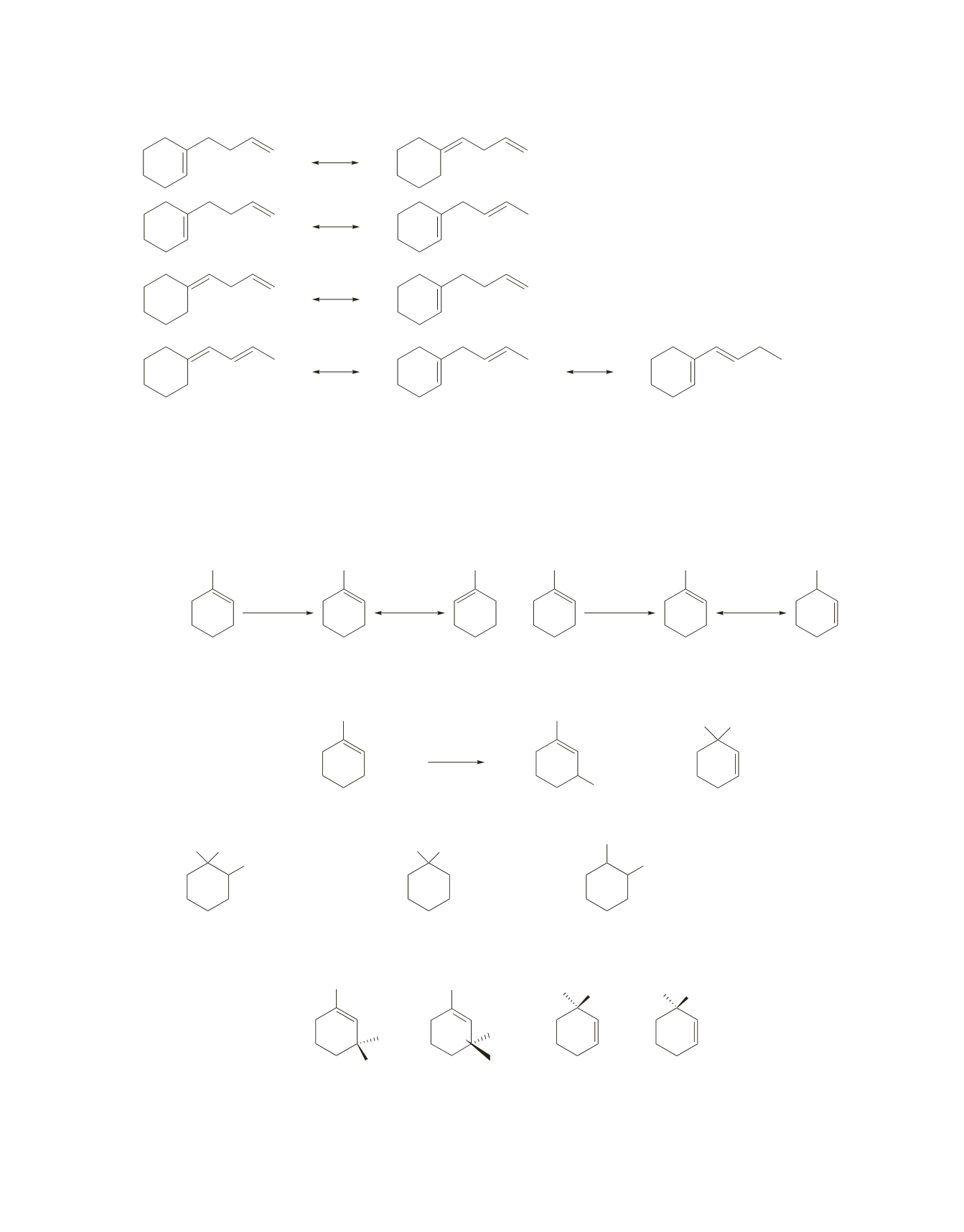
416 Chapter 12
Copyright © 2017 Pearson Education, Inc.
21.
a.
•
•
b.
•
•
c.
•
•
d.
•
•
•
22.
a. 1.
There are two sets of secondary allylic hydrogens,
a
and
b
(plus a less reactive set of primary allylic
hydrogens on the methyl group).
First, we need to determine which allylic hydrogen is the easiest one to remove. Removing one
of the
a
allylic hydrogens forms an intermediate in which the unpaired electron is shared by two
secondary allylic carbons. Removing one of the
b
allylic hydrogens forms an intermediate in which
the unpaired electron is shared by a tertiary allylic carbon and a secondary allylic carbon.
CH
3
removing an
H from
a
secondary
secondary
removing an
H from
b
secondary
CH
3
CH
3
CH
3
CH
3
CH
3
tertiary
a
b
•
•
•
•
Therefore, the major products are obtained by removing a
b
allylic hydrogen.
CH
3
a
b
1-methylcyclohexene
NBS,
peroxide
CH
3
Br
+
H
3
C Br
2.
H
3
C Br
Br
3.
H
3
C Br
4.
CH
3
Br
b. 1.
Each of the products has one asymmetric center.
The
R
and the
S
stereoisomers are obtained for each product.
CH
3
Br
H
CH
3
Br
Br
H
3
C
H
CH
3
Br
















