
422 Chapter 12
Copyright © 2017 Pearson Education, Inc.
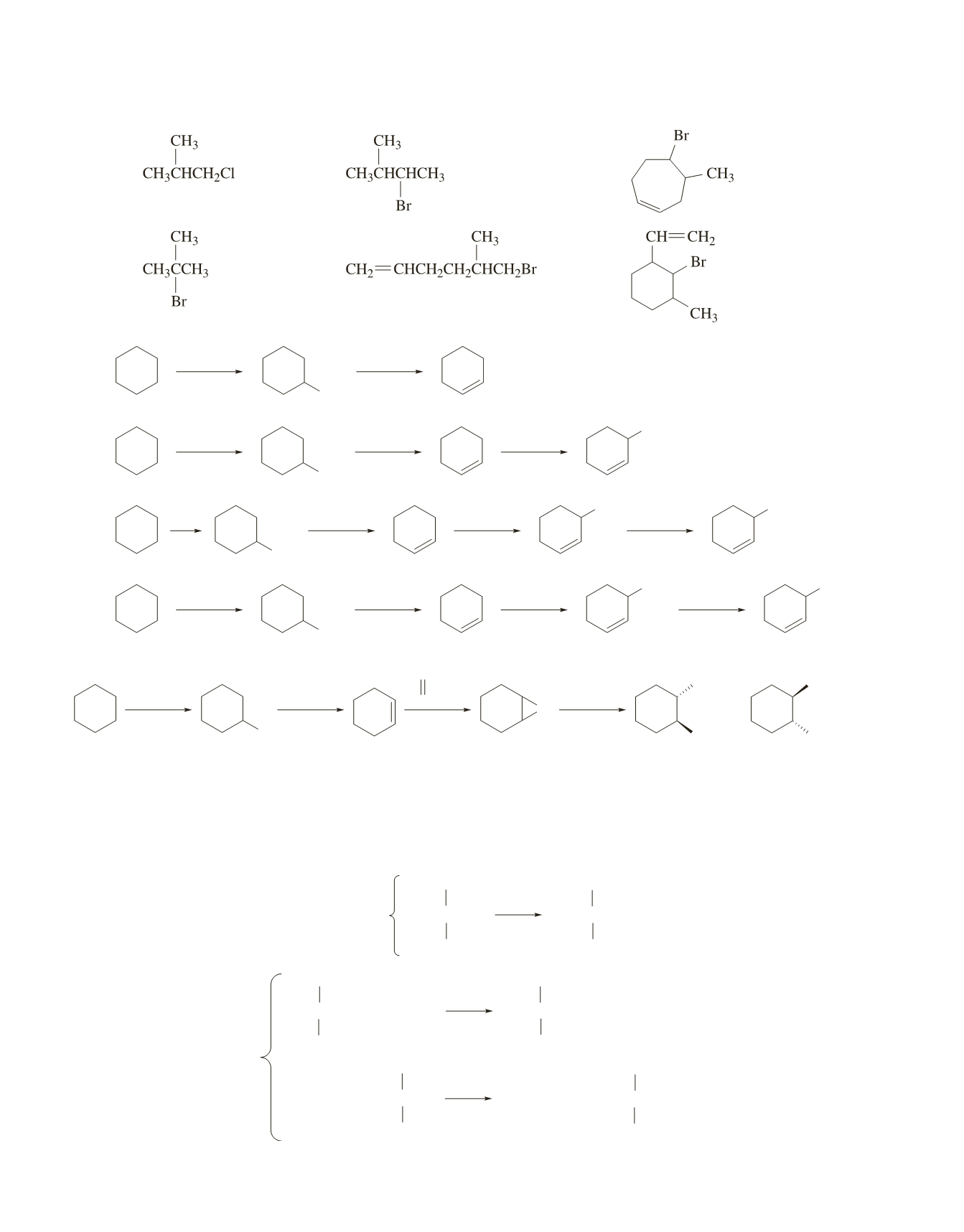
39.
a.
c.
e.
b.
d.
f.
40. a.
Br
Br
2
hv
tert
-BuO
−
b.
Br
Br
Br
2
hv
tert
-BuO
−
NBS,
peroxide
c.
Br
Br
OCH
3
Br
2
hv
tert
-BuO
−
CH
3
O
−
NBS,
peroxide
d.
Br
Br
CH
3
Br
2
hv
tert
-BuO
−
(CH
3
)
2
CuLi
NBS,
peroxide
e.
tert
-BuO
−
RCOOH
O
Br
2
hv
1. CH
3
O
−
2. HCl
Br
O
OCH
3
OH
OH
OCH
3
+
41.
a.
This reaction has two sets of propagation steps because two different radicals are generated in the
initiation step. This reaction also has several more termination steps because of the two different
radicals generated in the initiation step. (Bond dissociation enthalpies can be found in Table 5.1 on
page 206 of the text.)
CH
3
COCl
CH
3
CO
CH
3
CH
3
•
+
Cl
•
initiation
CH
3
CH
3
CH
3
COH
CH
3
CH
3
CH
3
CO
CH
3
CH
3
•
+
propagation
CH
3
CH
3
+
CH
3
CH
2
•
CH
3
CH
2
Cl
CH
3
COCl
CH
3
CH
3
+
+
CH
3
CH
2
•
CH
3
CO
CH
3
CH
3
•
















