
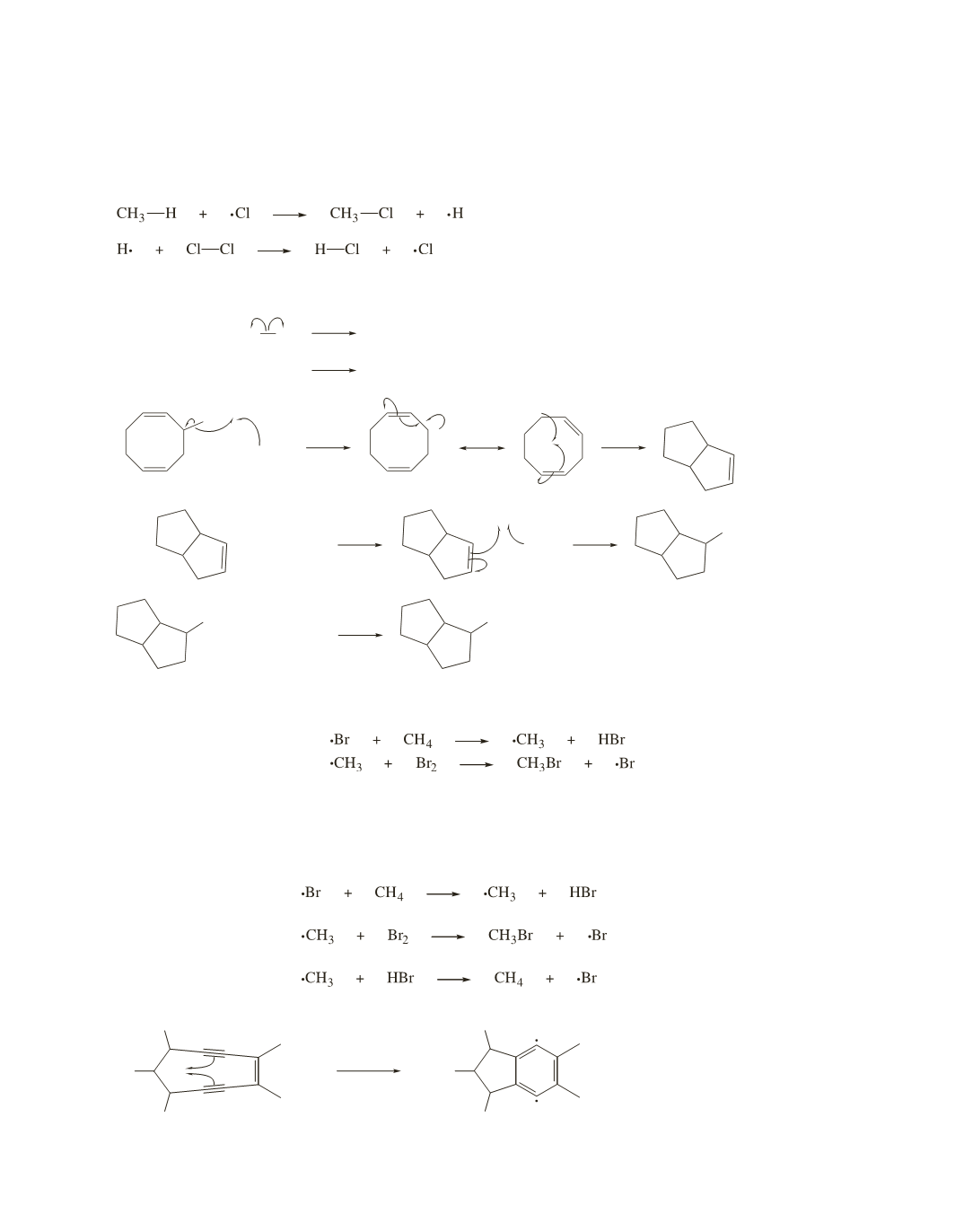
426 Chapter 12
Copyright © 2017 Pearson Education, Inc.
48.
The overall
∆
H
°
value is the same for both mechanisms. However, the mechanism that occurs is controlled
by the first propagation step. The first propagation step of the alternative mechanism is very endothermic,
so it would not be able to compete with the first propagation step of the mechanism shown in Problem 47b.
∆
H
°
=
105
-
84
=
21 kcal
>
mol
∆
H
°
=
58
-
103
=
-
45 kcal
>
mol
Overall
∆
H
°
=
21
+
1
-
45
2
=
-
24 kcal
>
mol
49.
RO OR
2 RO
•
CCl
3
•
RO
•
+
HCCl
3
+
H
•
•
•
•
HCCl
3
CCl
3
•
+
CCl
3
•
•
CCl
3
•
CCl
3
HCCl
3
CCl
3
+
CCl
3
•
ROH
+
+
+
50.
The methyl radical that is created in the first propagation step reacts with Br
2
, forming bromomethane.
If HBr is added to the reaction mixture, the methyl radical that is created in the first propagation step of the
bomination of methane can react with Br
2
or with the added HBr. Because only reaction with Br
2
forms
bromomethane (reaction with HBr re-forms methane), the overall rate of formation of bromomethane is
decreased.
51.
R
3
R
2
R
4
R
5
R
1
R
4
R
5
R
1
R
3
R
2
















