
856 Chapter 28
Copyright © 2017 Pearson Education, Inc.
30.
a.
This is a [1,5] sigmatropic rearrangement, so it can take place by a concerted mechanism under ther-
mal conditions.
H
H
C
H
C
CH
2
H
H
C
C
CH
3
b.
This is a [1,3] sigmatropic rearrangement, so it can take place by a concerted mechanism under photo-
chemical conditions.
CH
2
H
hv
CH
3
31.
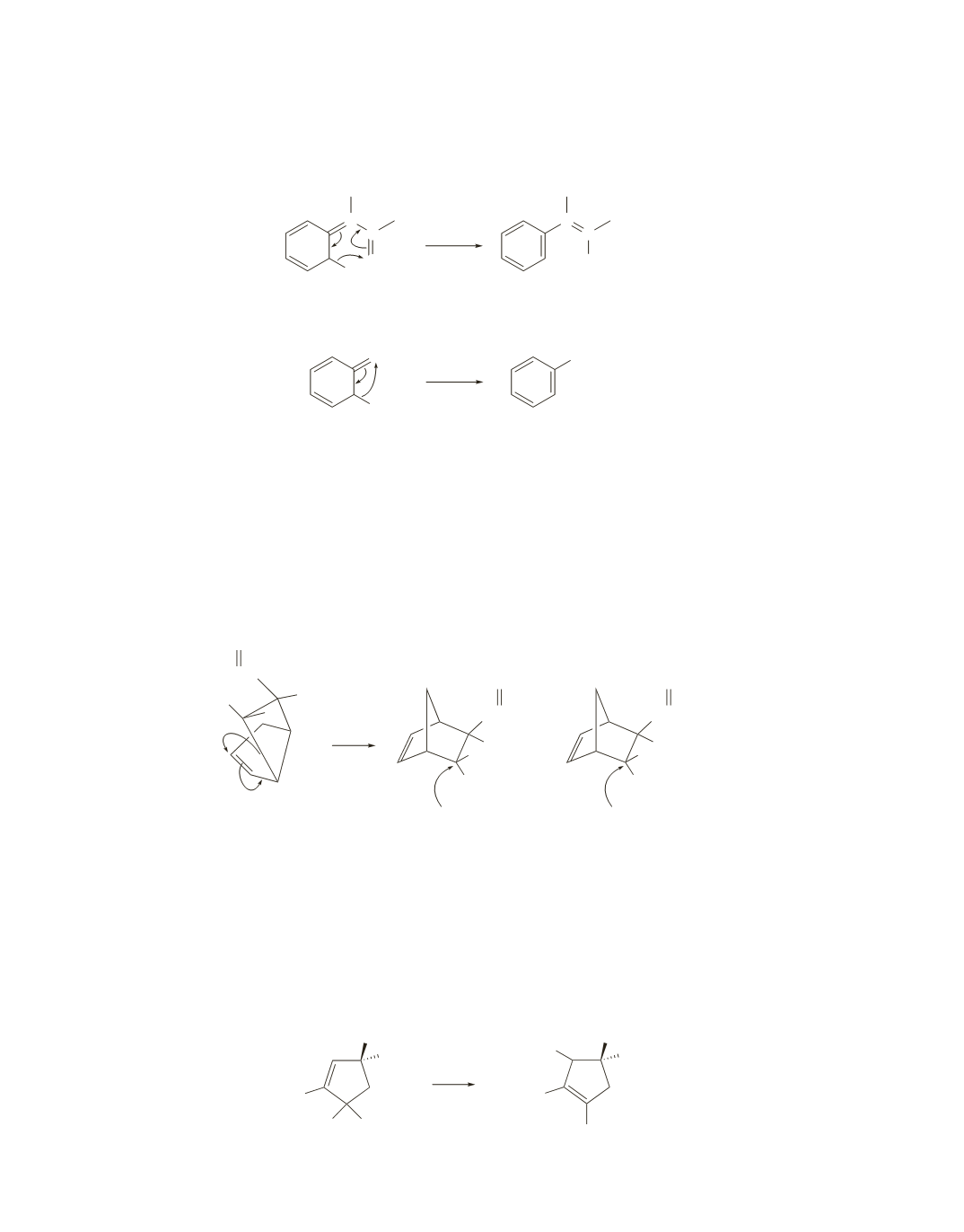
At first glance, it is surprising that the isomerization of Dewar benzene (a highly-strained and unstable
molecule) to benzene (a stable aromatic compound) is so slow. However, the isomerization requires
conrotatory ring opening, which is symmetry forbidden under thermal conditions. The reaction, therefore,
cannot take place by a concerted pathway and must take place by a much slower stepwise process.
32.
B
is the product. Because the reaction is a [1,3] sigmatropic rearrangement, antarafacial ring closure is
required. Carbon, therefore, must migrate using both lobes of its
p
orbital. This means that the configura-
tion of the migrating carbon will undergo inversion. The configuration of the migrating carbon has been
inverted in
B
(the H’s are cis to each other but were trans to each other in the reactant) and retained in
A
.
H
D
H
CO
O
H
D
H
OCCH
3
O
H
H
D
OCCH
3
CH
3
O
the H’s are trans
to each other
A
B
retention
inversion
the H’s are trans
to each other
the H’s are cis
to each other
33.
Hydrogen cannot undergo a [1,3] sigmatropic rearrangement, because it cannot migrate by an antarafacial
pathway that is required for a sigmatropic rearrangement involving an even number of pairs of electrons
under thermal conditions. Carbon can undergo a [1,3] sigmatropic rearrangement because it can migrate by
a suprafacial pathway if it uses both lobes of its
p
orbital. Therefore, the first compound can undergo only
a 1,3-methyl group migration.
H
3
C
H
CH
3
H
3
C
H
3
C
CH
3
H
3
C
H
H
H
















