
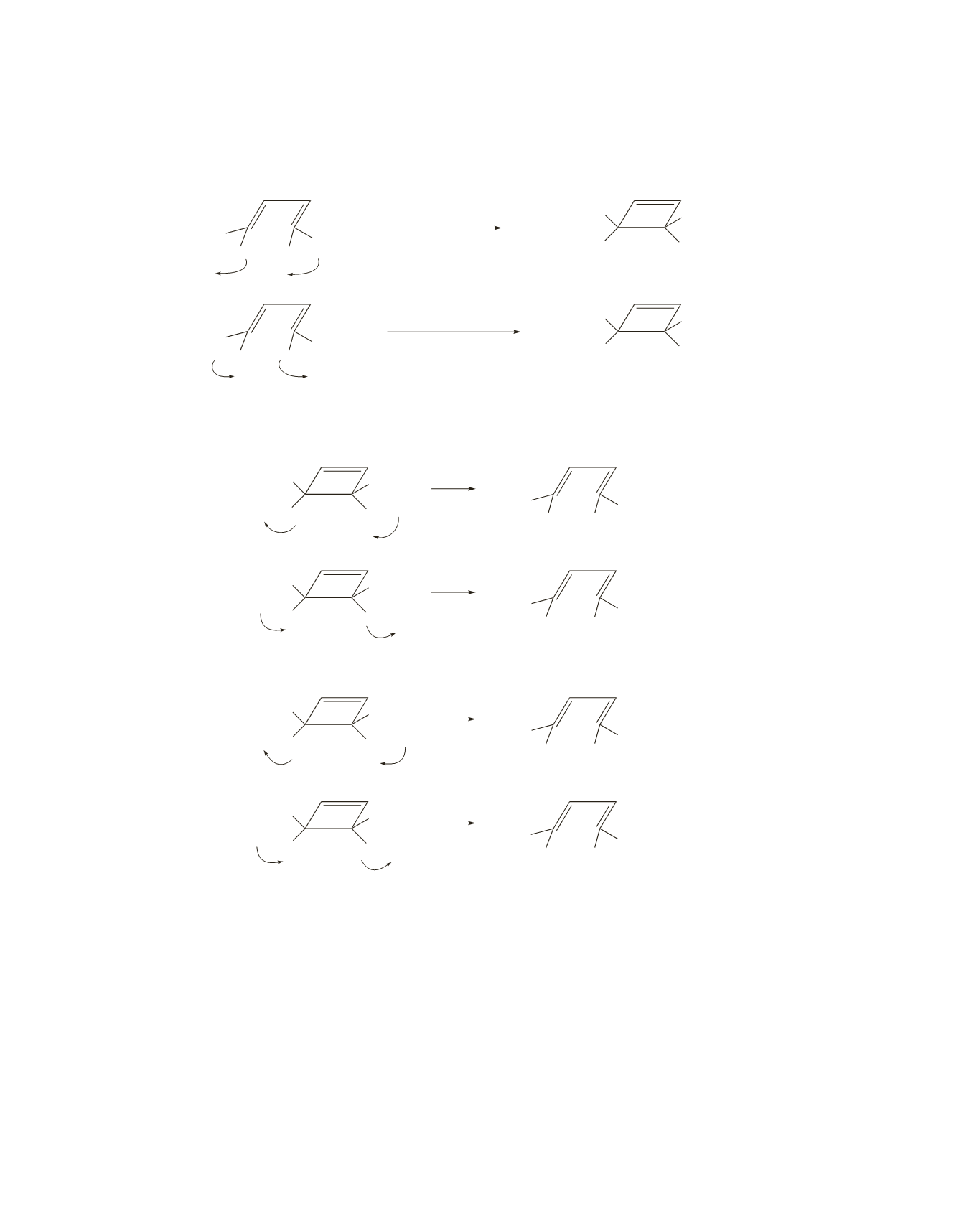
860 Chapter 28
Copyright © 2017 Pearson Education, Inc.
41.
Because the reactant has two
p
bonds, electrocyclic ring closure is conrotatory. Two different
compounds,
X
and
Y
, can be formed because conrotatory ring closure can occur in either a clockwise or
counterclockwise direction.
C
6
H
5
C
6
H
5
CD
3
CH
3
CD
3
C
6
H
5
C
6
H
5
CH
3
C
6
H
5
C
6
H
5
CD
3
C
6
H
5
C
6
H
5
CD
3
CH
3
both clockwise
both counterclockwise
Y
X
CH
3
Each of the compounds (
X
and
Y
) can undergo a conrotatory ring-opening reaction in either a clockwise
or counterclockwise direction to form either
A
or
B
.
A
and
B
are the only isomers that can be formed;
formation of
C
and
D
requires disrotatory ring closure.
CH
3
CD
3
C
6
H
5
C
6
H
5
C
6
H
5
CD
3
CH
3
C
6
H
5
X
B
CH
3
CD
3
C
6
H
5
C
6
H
5
CH
3
C
6
H
5
C
6
H
5
CD
3
X
A
C
6
H
5
C
6
H
5
CD
3
CH
3
CH
3
C
6
H
5
C
6
H
5
CD
3
Y
A
C
6
H
5
C
6
H
5
CD
3
CH
3
C
6
H
5
CD
3
CH
3
C
5
H
5
Y
B
















