
Chapter 28 861
Copyright © 2017 Pearson Education, Inc.
42.
H
3
C
O
CH
3
H
3
C
O
CH
3
H
3
C
O
CH
3
H
H
3
C
OH
CH
3
Claisen
rearrangement
Cope
rearrangement
tautomerization
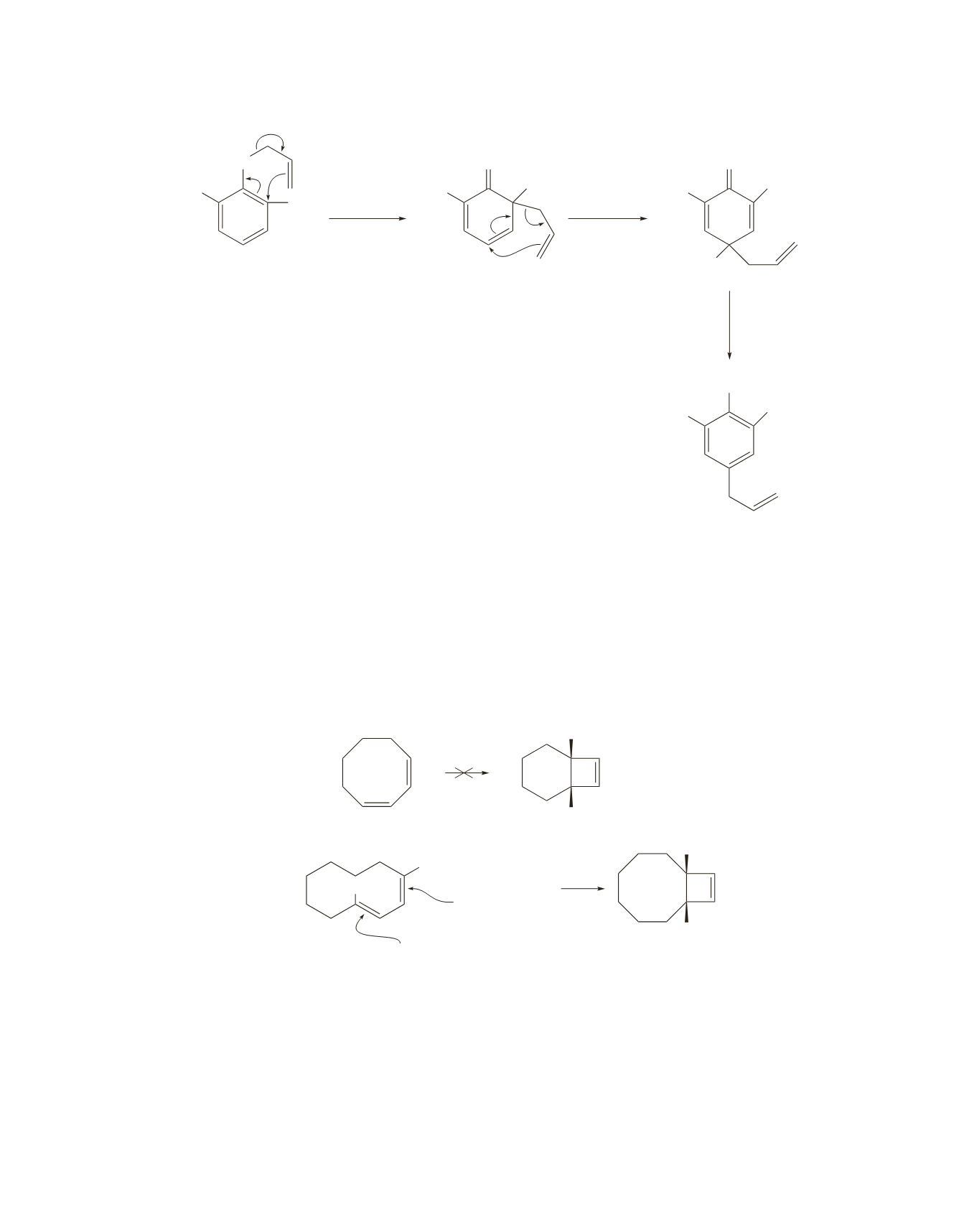
43.
Because the compounds that undergo ring closure to give
A
and
B
have two
p
bonds, ring opening of
A
and
B
under thermal conditions is conrotatory. Because the hydrogens in
A
and
B
are cis, they must point
in the same direction in the ring-opened product. To have the two hydrogens pointing in the same direction,
one of the double bonds in the ring-opened compound must be cis and the other must be trans.
An eight-membered ring is too small to accommodate conjugated double bonds with one cis and the other
trans, so
A
will not be able to undergo a ring-opening reaction under thermal conditions. A 10-membered
ring can accommodate a trans double bond, so
B
is able to undergo a ring-opening reaction under thermal
conditions.
H
H
H
H
H
H
Both double bonds are cis.
trans double bond
cis double bond
A
B
















