
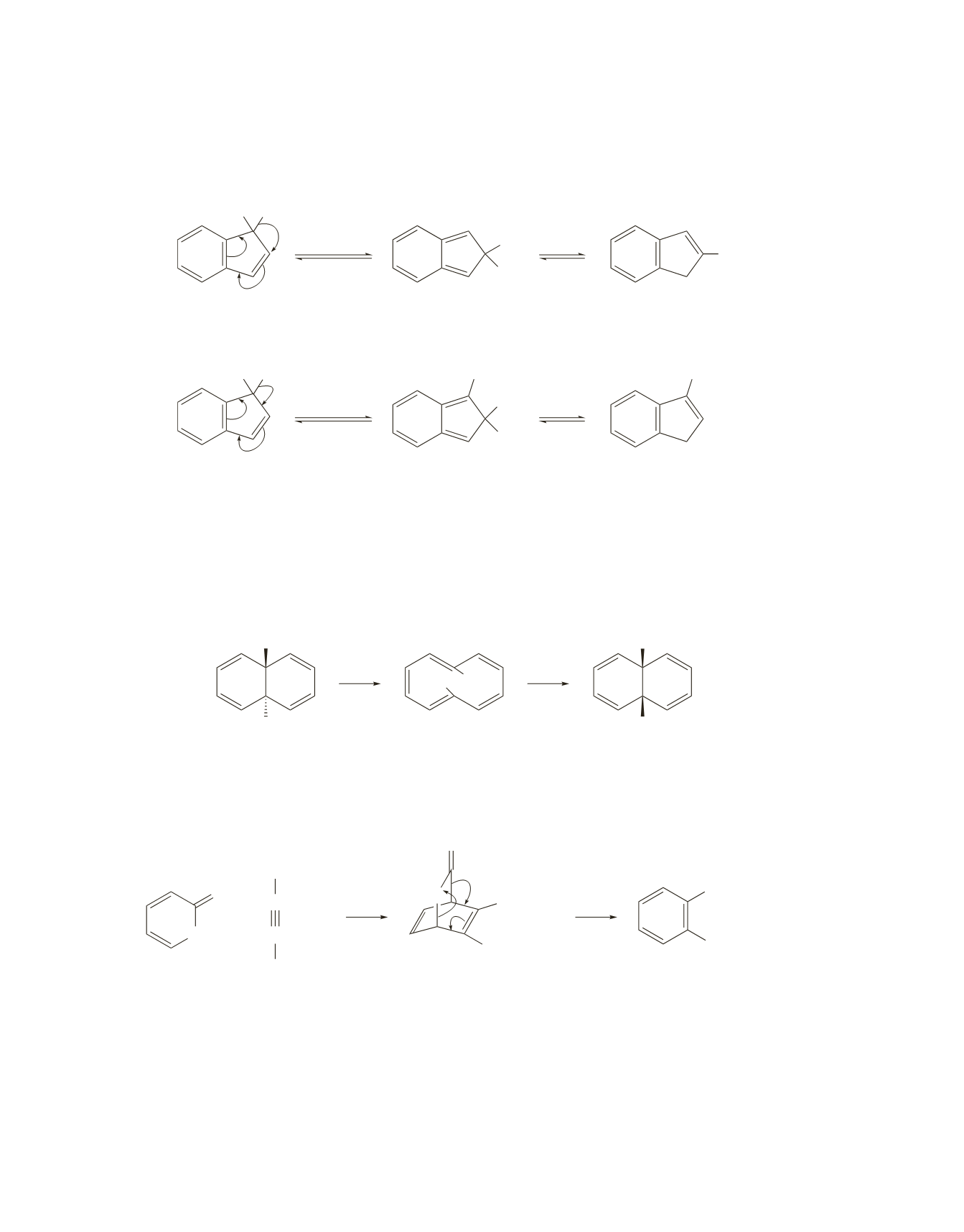
862 Chapter 28
Copyright © 2017 Pearson Education, Inc.
44.
The compound undergoes a 1,5-hydrogen shift of D or a 1,5-hydrogen shift of H. In each case, an unstable
nonaromatic intermediate is formed that undergoes a subsequent 1,5-hydrogen shift to form an aromatic
product.
D
D
H
D
D
H
H
[1,5]
[1,5]-shift
[1,5]
[1,5]-shift
nonaromatic
aromatic
aromatic
nonaromatic
DH
HD
45.
Because the ring-opened compound formed in the first step has three conjugated
p
bonds involved in an
electrocyclic reaction, conrotatory ring opening of the reactant will occur under photochemical conditions,
and the trans hydrogens in the reactant require that the hydrogens point in the opposite direction in the
ring-opened compound. Thermal electrocyclic ring closure of a three
p
bond system is disrotatory in step
two, and disrotatory ring closure of a compound with hydrogens that point in opposite directions will cause
those hydrogens to be cis in the ring-closed product.
H
H
H
H
H H
hv
46.
A Diels–Alder reaction is followed by a reverse Diels–Alder reaction that eliminates CO
2
. Loss of a
stable gas molecule
1
CO
2
2
and formation of a stable aromatic product provide the driving force for the
second step.
O
O
C
C
CO
2
CH
3
CO
2
CH
3
+
O
O
CO
2
CH
3
CO
2
CH
3
CO
2
CH
3
CO
2
CH
3
+
CO
2
















