
Chapter 28 859
Copyright © 2017 Pearson Education, Inc.
38.
a.
CH
2
CH
2
CH
2
CH
2
b.
CH
2
CH
2
CH
3
CH
3
CH
3
CH
3
+
C
C
CH
3
H
H H
3
C
39.
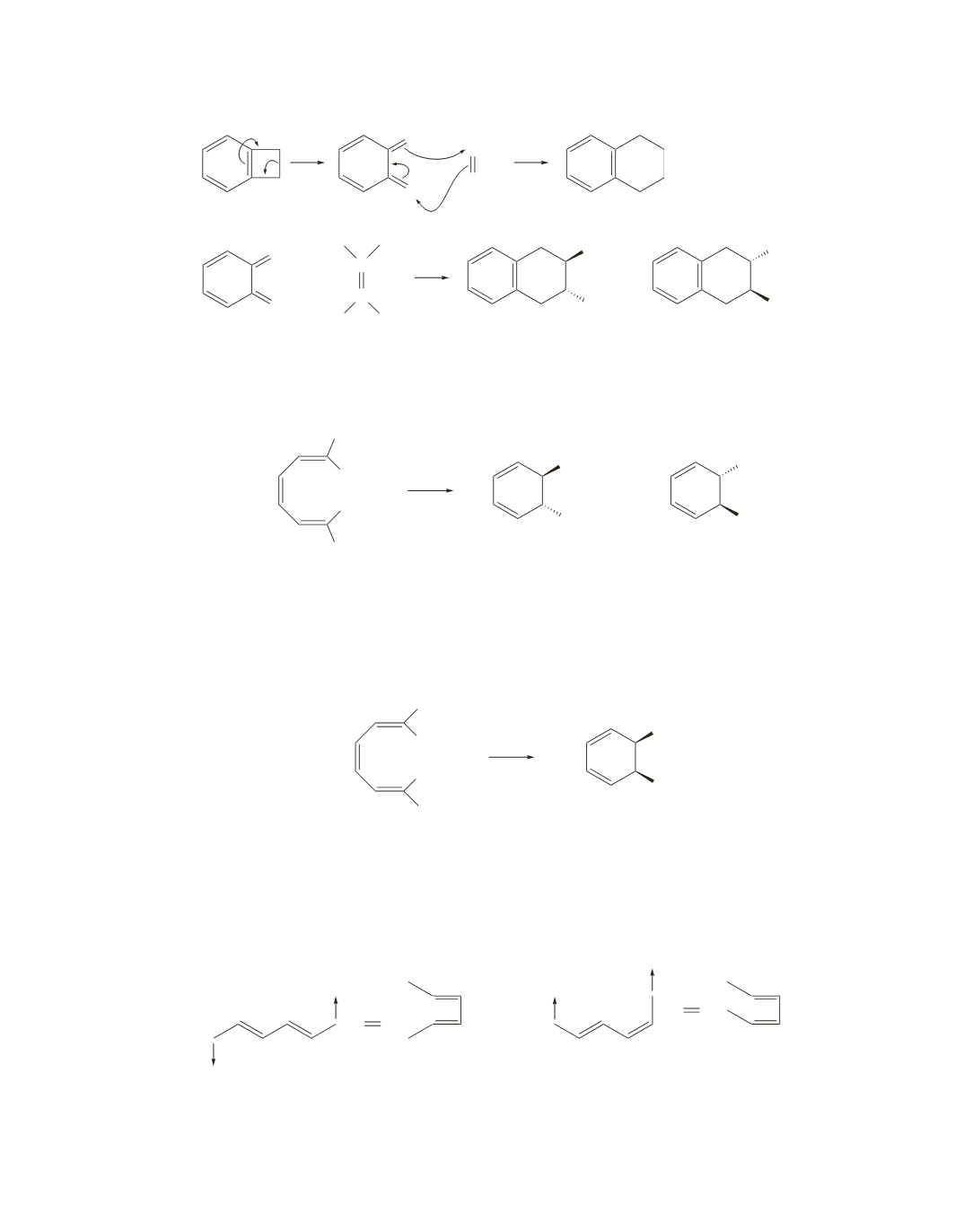
Disrotatory ring closure of (2
E
,4
Z
,6
Z
)-octatriene leads to the trans isomer, which can exist as a pair of
enantiomers. One enantiomer is formed if the “top lobes” of the
p
orbitals rotate toward each other, and the
other enantiomer is formed if the “bottom lobes” of the
p
orbitals rotate toward each other.
CH
3
H
CH
3
H
CH
3
CH
3
+
CH
3
CH
3
(2
E
,
4
Z
,
6
Z
)-octatriene
In contrast, disrotatory ring closure of (2
E
,4
Z
,6
E
)-octatriene leads to the cis isomer, which is a meso
compound and, consequently, does not have a nonsuperimposable mirror image. Therefore, the same
compound is formed from the “top lobes” of the
p
orbitals rotating toward each other and from the “bottom
lobes” of the
p
orbitals rotating toward each other.
H
H
CH
3
CH
3
CH
3
CH
3
(2
E
,
4
Z
,
6
E
)-octatriene
40.
Under thermal conditions, a compound with two
p
bonds undergoes conrotatory ring closure. Conrotatory
ring closure that results in a ring-closed compound with the substituents cis to each other requires that the
substituents point in the same direction in the reactant. Therefore, the product with the methyl substituents
pointing in the same direction is obtained in 99% yield.
1%
Methyl groups point in opposite directions.
99%
Methyl groups point in the same directions.
















