
854 Chapter 28
Copyright © 2017 Pearson Education, Inc.
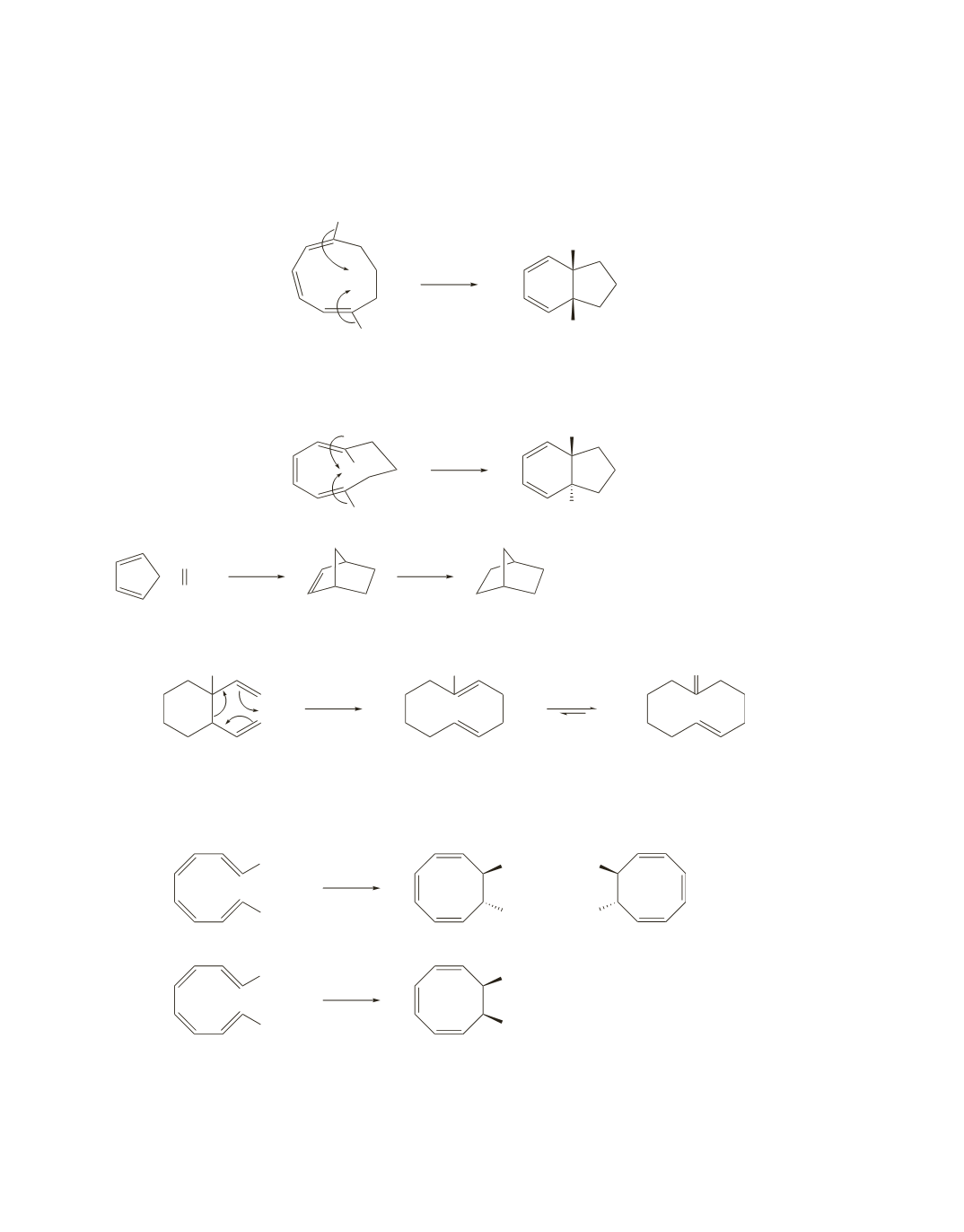
25.
The hydrogens that end up at the ring juncture in the first reaction point in opposite directions in the
reactant. Because ring closure is disrotatory (odd number of
p
bonds, thermal conditions), the hydrogens
in the ring-closed product are cis. (See Table 28.2 on page 1224 of the text.)
H
H
H
H
In the second example, the hydrogens that end up at the ring juncture point in the same direction in the
reactant. Ring closure is still disrotatory, so the hydrogens in the ring-closed product are trans.
H
H
H
H
26.
CH
2
CH
2
H
2
Pd/C
27.
The first step is similar to a Cope rearrangement; the second step is tautomerization of the enol.
OH
OH
O
28.
1.
Because the compound has an even number of
p
bonds, it undergoes conrotatory ring closure under
thermal conditions and disrotatory ring closure under photochemical conditions. Because the two
methyl substituents point in opposite directions, they will be trans in the ring-closed product when ring
closure is conrotatory and cis in the ring-closed product when ring closure is disrotatory.
a.
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
hv
H
3
C
H
3
C
+
b.
2.
Because the compound has an even number of
p
bonds, it will undergo conrotatory ring closure under
thermal conditions and disrotatory ring closure under photochemical conditions. Because the two
methyl substituents point in the same direction, they will be cis in the ring-closed product when ring
closure is conrotatory and trans in the ring-closed product when ring closure is disrotatory.
















