
852 Chapter 28
Copyright © 2017 Pearson Education, Inc.
16.
Solved in the text.
17.
[1,3] Sigmatropic migrations of hydrogen cannot occur under thermal conditions because the four-membered transition state does not allow the required antarafacial rearrangement.
[1,3] Sigmatropic migrations of carbon can occur under thermal conditions because carbon can achieve the
required antarafacial rearrangement by using both lobes of its
p
orbital when it migrates.
18.
a.
Because 1,3-migration of carbon requires carbon to migrate using both lobes of its
p
orbital (it involves
an even number of pairs of electrons, so it takes place by an antarafacial pathway), migration occurs
with inversion of configuration.
b.
Because 1,5-migration of carbon requires carbon to migrate using only one lobe of its
p
orbital (it involves
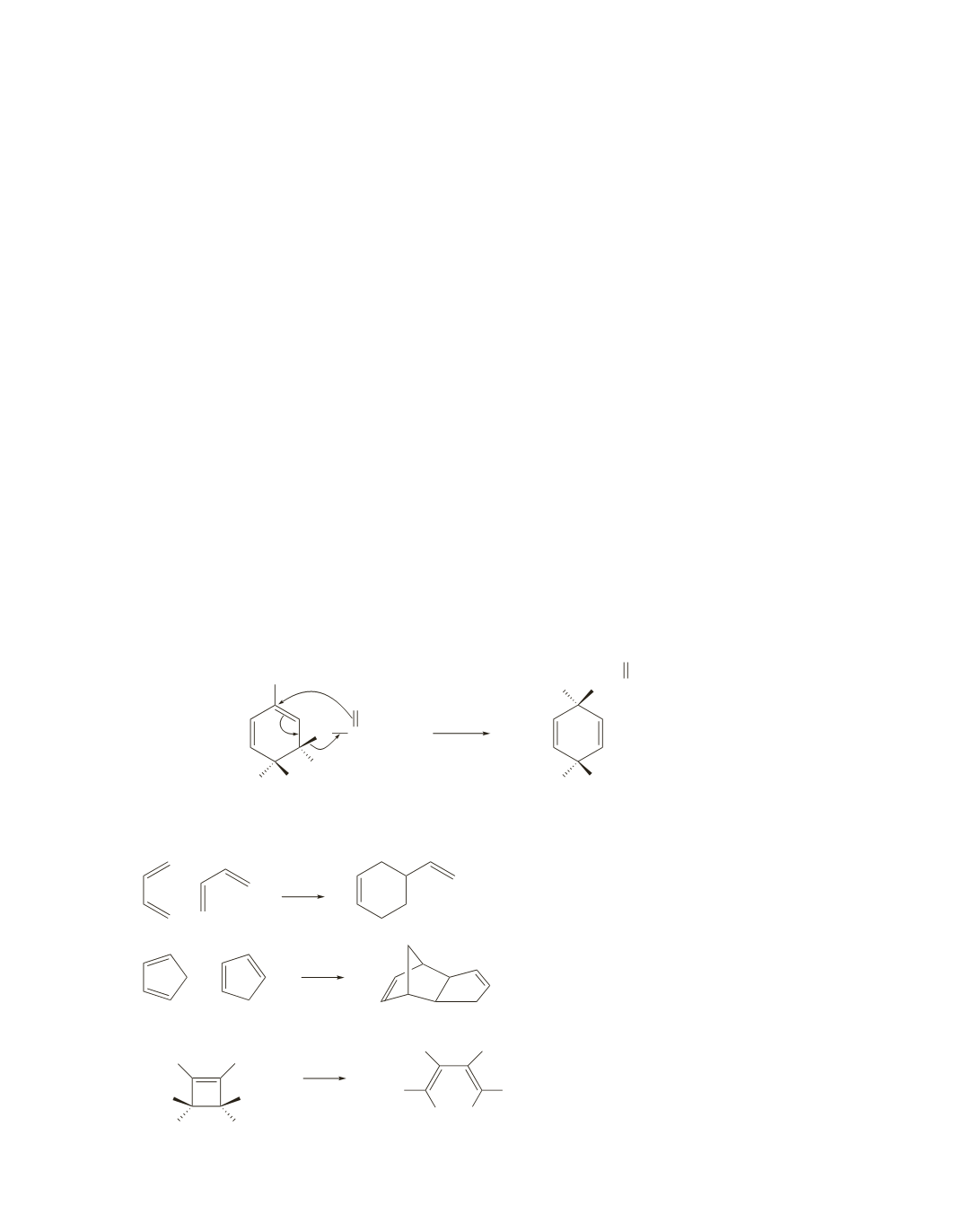
an odd number of pairs of electrons, so it takes place by a suprafacial pathway), migration occurs with
retention of configuration.
19.
Because the [1,7] sigmatropic rearrangement takes place under thermal conditions and involves an even
number (4) of pairs of electrons, migration of hydrogen involves antarafacial rearrangement. Because the
cyclic transition state involves eight ring atoms, antarafacial rearrangement is possible.
20.
Because the reactant (provitamin D
3
) has an odd number (3) of conjugated
p
bonds and reacts under
photochemical conditions, ring closure is conrotatory. The methyl and hydrogen substituents point in
opposite directions in provitamin D
3
. Conrotatory ring closure causes substituents that point in opposite
directions in the reactant to be trans in the product.
21.
Chorismate mutase catalyzes a [3,3] sigmatropic Claisen rearrangement.
COO
−
O
H HO
CCOO
−
CH
2
−
OOC CH
2
CCOO
−
H HO
O
H
22.
Were you able to convince yourself that TE-AC is valid?
23.
a.
H
3
C
H
3
C
CH
3
CH
3
H
H
H
3
C
H
3
C
CH
3
CH
3
H
H
b.
c.
















