
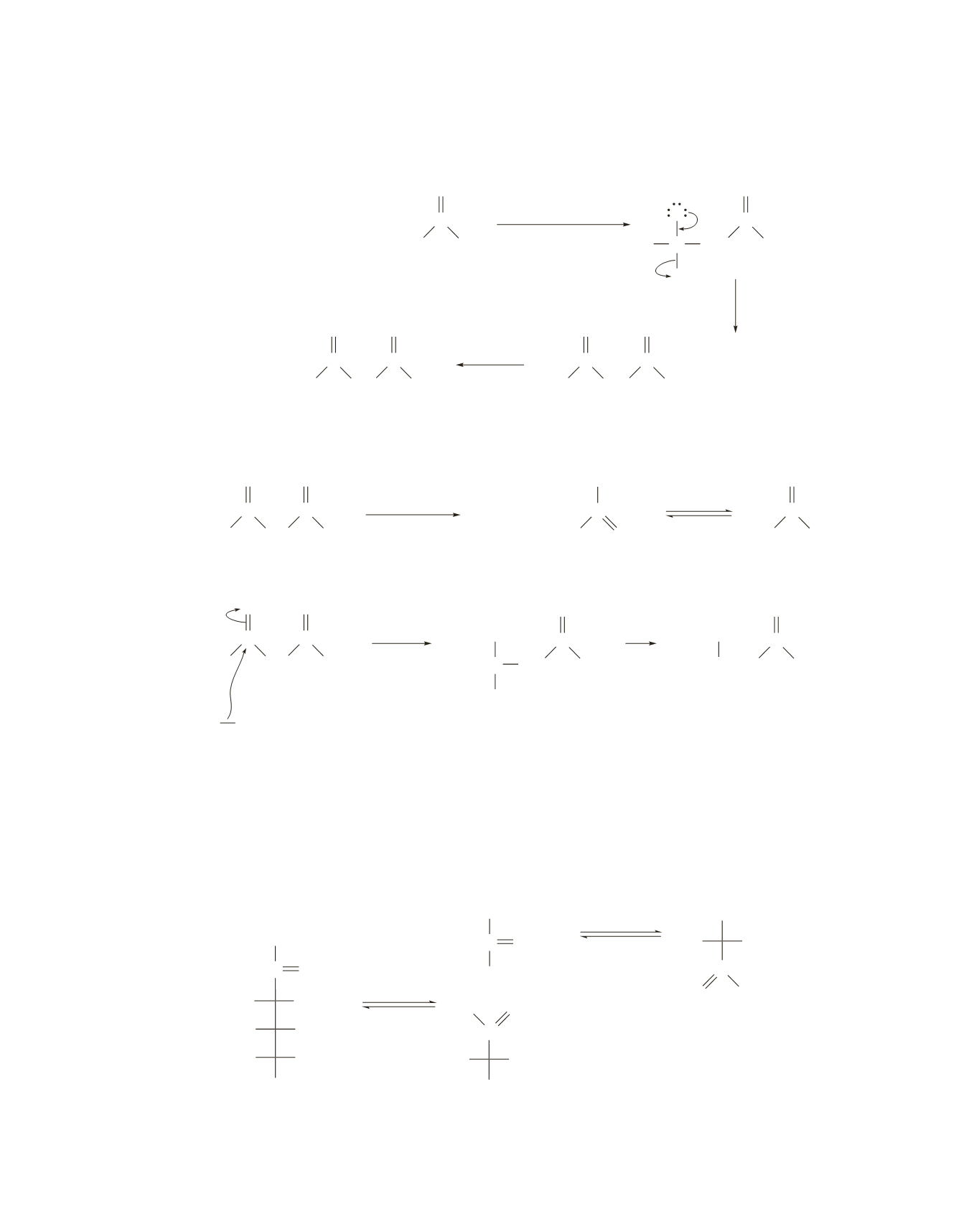
Chapter 24 787
Copyright © 2017 Pearson Education, Inc.
50.
A Claisen condensation between two molecules of acetyl-CoA forms acetoacetyl-CoA that, when hydro-
lyzed, forms acetoacetate.
CH
2
SCoA
C
+
Claisen condensation
CH
3
O
SCoA
CH
3
SCoA
acetyl-CoA
CH
3
CH
2
SCoA
hydrolysis
CH
3
CH
2
O
−
acetoacetyl-CoA
acetoacetate
+
CoASH
CoASH
−
C
O
C
O
C
O
C
O
C
O
C
O
Acetoacetate can undergo decarboxylation to form acetone, or it can be reduced to 3-hydroxybutyrate.
CH
2
O
−
O
−
CH
3
C
enolization
H
acetone
CH
3
CH
2
O
−
acetoacetate
+
CH
3
CH
2
decarboxylation
CO
2
acetoacetate
reduction
H
+
H
+
CH
3
CHCH
2
O
−
3-hydroxybutyrat
e
OH
NAD H
NAD
+
C
O
CH
3
CH
3
C
O
C
O
C
O
C
O
CH
3
CH
2
O
−
C
O
C
O
C
OH
51.
The mechanism for the conversion of fructose-1,6-bisphosphate to glyceraldehyde-3-phosphate and
dihydroxyacetone phosphate is shown on page 1058 of the text.
The mechanism for the conversion of dihydroxyacetone phosphate to glyceraldehyde-3-phosphate is
shown on page 1109 of the text.
From these mechanisms, you can see that the label (*) was at C-1 in glyceraldehyde-3-phosphate.
*
CH
2
OPO
3
2
−
OH
H
C
O H
*
H
HO
C
O
H
C
CH
2
OPO
3
2
−
H HO
OH H
OH H
O
*
*
CH
2
OPO
3
2
−
C
CH
2
OH
O
CH
2
OPO
3
2
−
CH
2
OPO
3
2
−
*
aldolase
see page 1058
of the text
triosephosphate
isomerase
see page 1109
of the text
















