
Chapter 19 661
Copyright © 2017 Pearson Education, Inc.
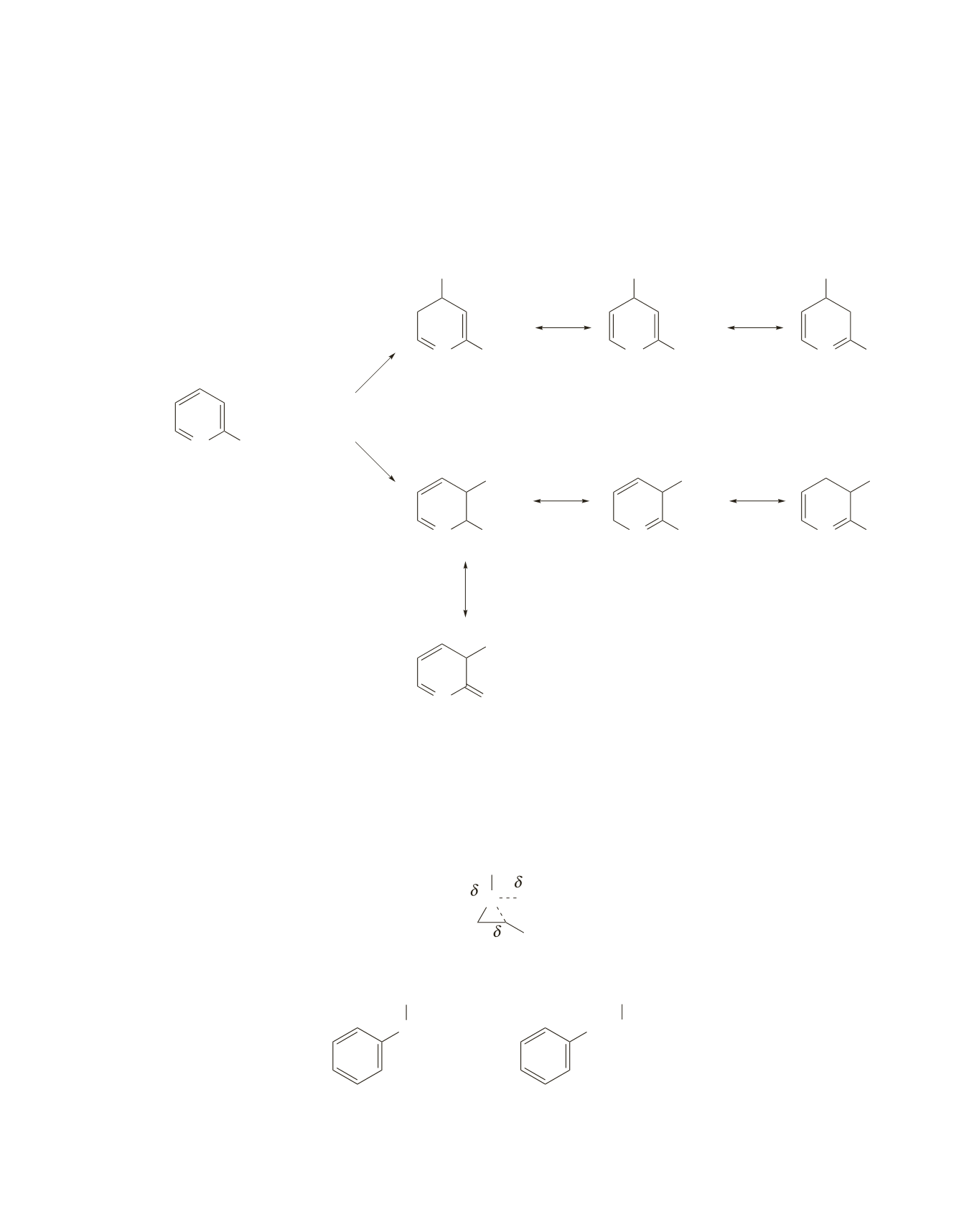
26.
By comparing the carbocation intermediates formed in the rate-determining step when the amino-substituted
compound undergoes electrophilic substitution at C-3 and C-4, you can see that the carbocation formed in
the case of substitution at C-3 is more stable; one of its resonance contributors is relatively stable because it
is the only one that does not have an atom with an incomplete octet. As a result, the amino-substituted com-
pound undergoes electrophilic substitution predominantly at C-3. Therefore, the keto-substituted compound
is the one that undergoes electrophilic substitution predominantly at C-4.
N NHR
+
Y
+
N
substitution at C-4
substitution at C-3
NHR
Y
+
N NHR
+
Y
N NHR
Y
+
N NHR
Y
+
N NHR
+
N NHR
Y
Y
+
N NHR
+
Y
relatively stable
27.
a.
The Lewis acid, AlCl
3
, complexes with nitrogen, causing the aziridine ring to open when it is attacked
by the nucleophilic benzene ring. The ring will open in the direction that puts the partial positive
charge on the more substituted carbon because it is more stable than the transition state that would
have the positive charge on the less substituted carbon.
N AlCl
3
CH
3
CH
3
+
−
+
Therefore, the major and minor products are those shown.
CHCH
2
NHCH
3
CH
3
CH
2
CHNHCH
3
CH
3
major
minor
b.
Yes, epoxides can undergo similar reactions.
















