
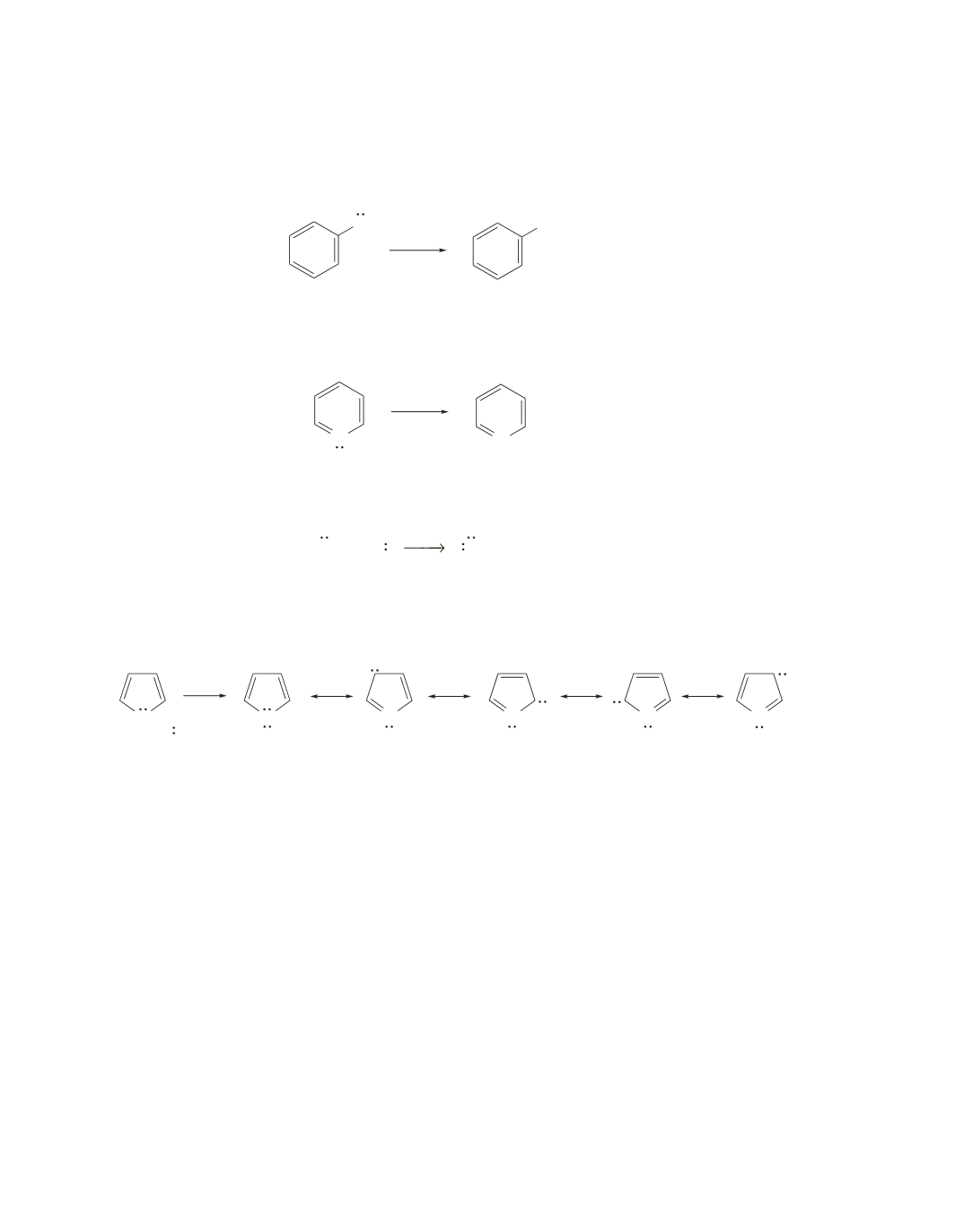
664 Chapter 19
Copyright © 2017 Pearson Education, Inc.
35.
A UV spectrum results from the
p
electron system. The lone-pair electrons on the nitrogen in aniline are
delocalized into the benzene ring and, therefore, are part of the
p
system. Protonation of aniline removes
two electrons from the
p
system. This has a significant effect on its UV spectrum.
NH
2
3
HCl
+
−
NH
Cl
+
The lone-pair electrons on the nitrogen atom in pyridine are
sp
2
electrons and thus are not part of the
p
system. Protonation of pyridine, therefore, does not remove any electrons from the
p
system and has only
a minor effect on the UV spectrum.
N
N
H
HCl
+
Cl
+
−
36.
When ammonia loses a proton, the electrons left behind remain on nitrogen.
N
N
H
H HB
3
2
− +
+
B
+
When pyrrole loses a proton, the electrons left behind can be delocalized onto the four ring carbons.
Electron delocalization stabilizes the anion and makes it easier to form. Recall that stabilizing the base
increases the acidity of its conjugate acid.
N
H
N
N
N
N
N
−
−
−
−
−
+
+
H
+
B
B
















