
Chapter 19 665
Copyright © 2017 Pearson Education, Inc.
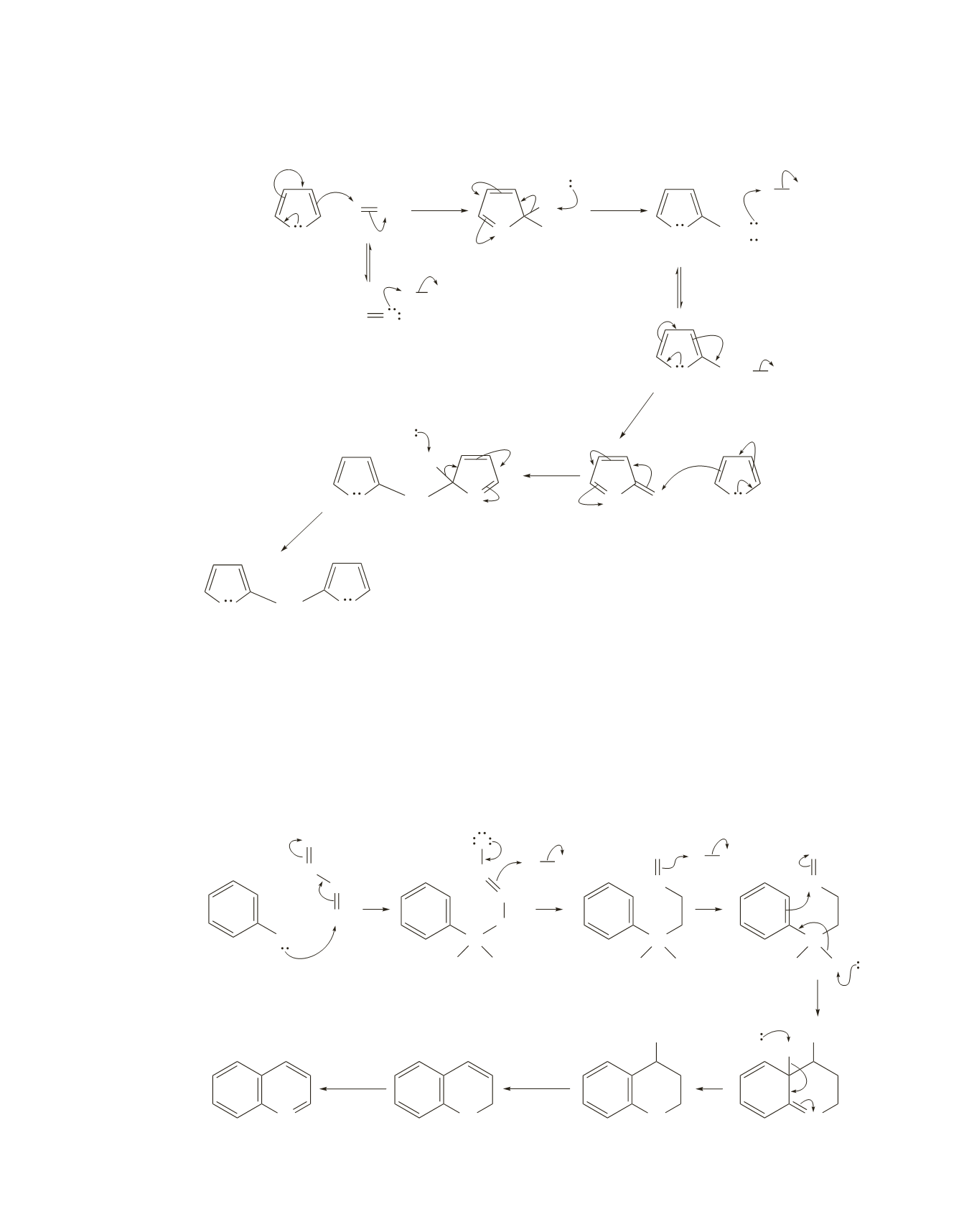
37.
The electrophile adds preferentially to the 2-position.
+
N
H
H
2
C OH
+
H
2
C O
H B
+
N
H
CH
2
OH
H
B
N
H
CH
2
OH
H B
+
N
H
CH
2
OH
H
+
N
H
CH
2
+
N
H
H
2
O
+
N
H
N
H
CH
2
N
H
N
H
CH
2
HB
+
+
H
B
+
38.
Before we can answer the questions, we must figure out the mechanism of the reaction. Once the mecha-
nism is known, it will be relatively easy to determine how a change in a reactant will affect the product.
The mechanism is shown below.
Propenal, an
a
,
b
-unsaturated aldehyde, undergoes a conjugate addition reaction with aniline. This is fol-
lowed by an intramolecular electrophilic aromatic substitution reaction. Dehydration of the alcohol results
in 1,2-dihydroquinoline, which is oxidized to quinoline by nitrobenzene.
NH
2
CH
2
CH
HC
O
H B
+
+
N
CH
2
CH
HC
O
H H
−
HC
O H B
+
+
N
H
OH
H
B
+
N
H
OH
N
H
+
H
2
O
oxidation
dehydration
with
C
6
H
5
NO
2
N
+
N
H H
HC
OH
+
N
H H B
















