
Chapter 19 667
Copyright © 2017 Pearson Education, Inc.
b.
The electrophile adds preferentially to the 2-position.
O
Br Br
O Br
+
CH
3
OH
O
CH
3
O
Br
O
CH
3
O
Br
H
+
O
CH
3
O
OCH
3
O
CH
3
O
OCH
3
H
+
B
B
+
O
CH
3
O
+
CH
3
OH
Br
−
40.
a.
O
O
HNO
3
O
2
N
CH
3
CH
3
C
O
C
O
You can see why the nitro substituent goes to this position by examining the relative stabilities of the pos-
sible carbocation intermediates.
O
O
Y
+
Y
+
O Y
+
O Y
+
O
Y
+
O
+
Y
O
+
Y
O
O
Y
+
+
O
+
O
+
Y
Y
Y
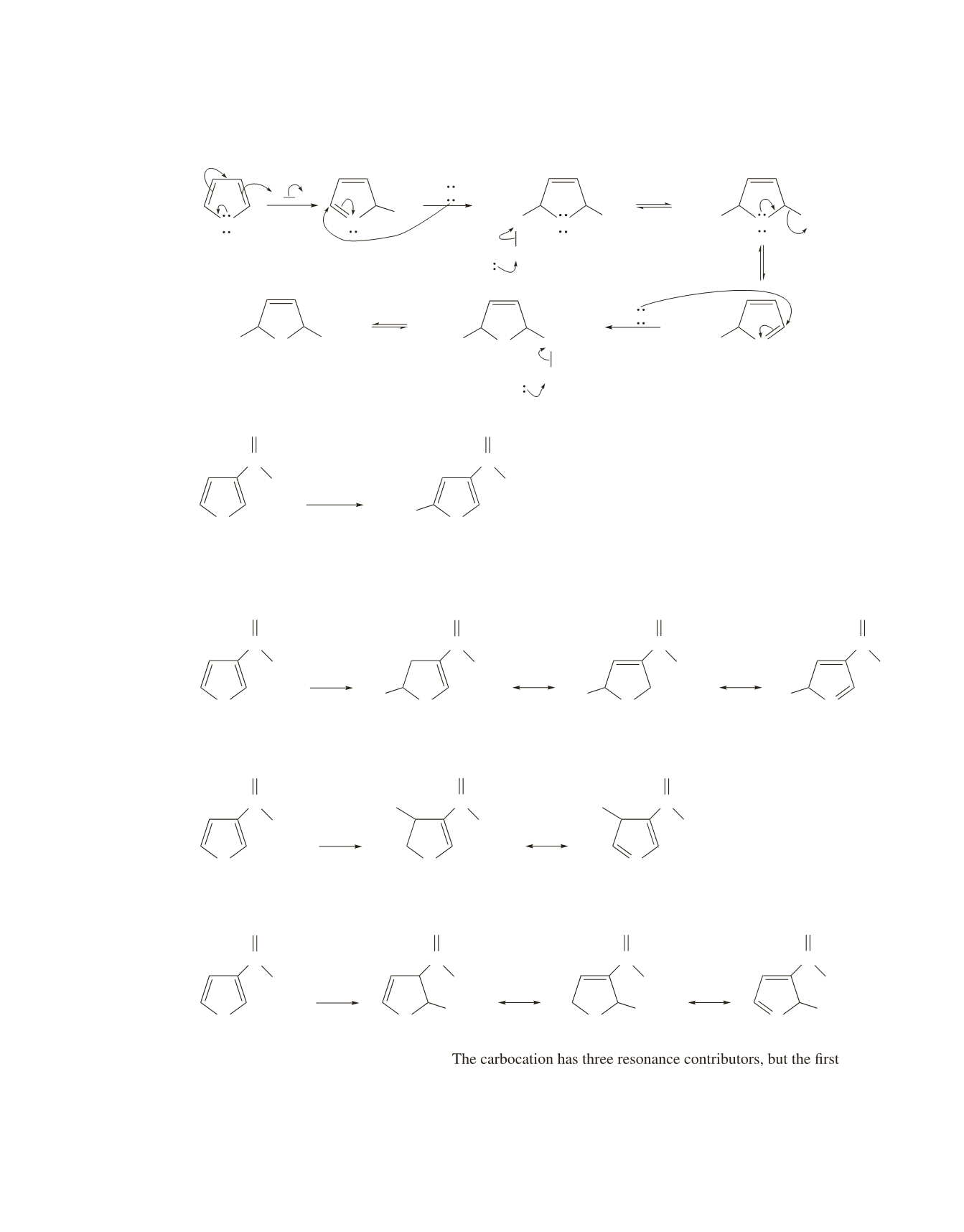
The carbocation has three resonance contributors.
The carbocation has two resonance contributors.
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
relatively
unstable
C
O
C
O
C
CH
3
O
C
O
C
O
C
O
C
O
C
O
C
O
C
O
C
O
one is relatively unstable because the positive charge is on
the carbon attached to the electron-withdrawing substituent.
















