
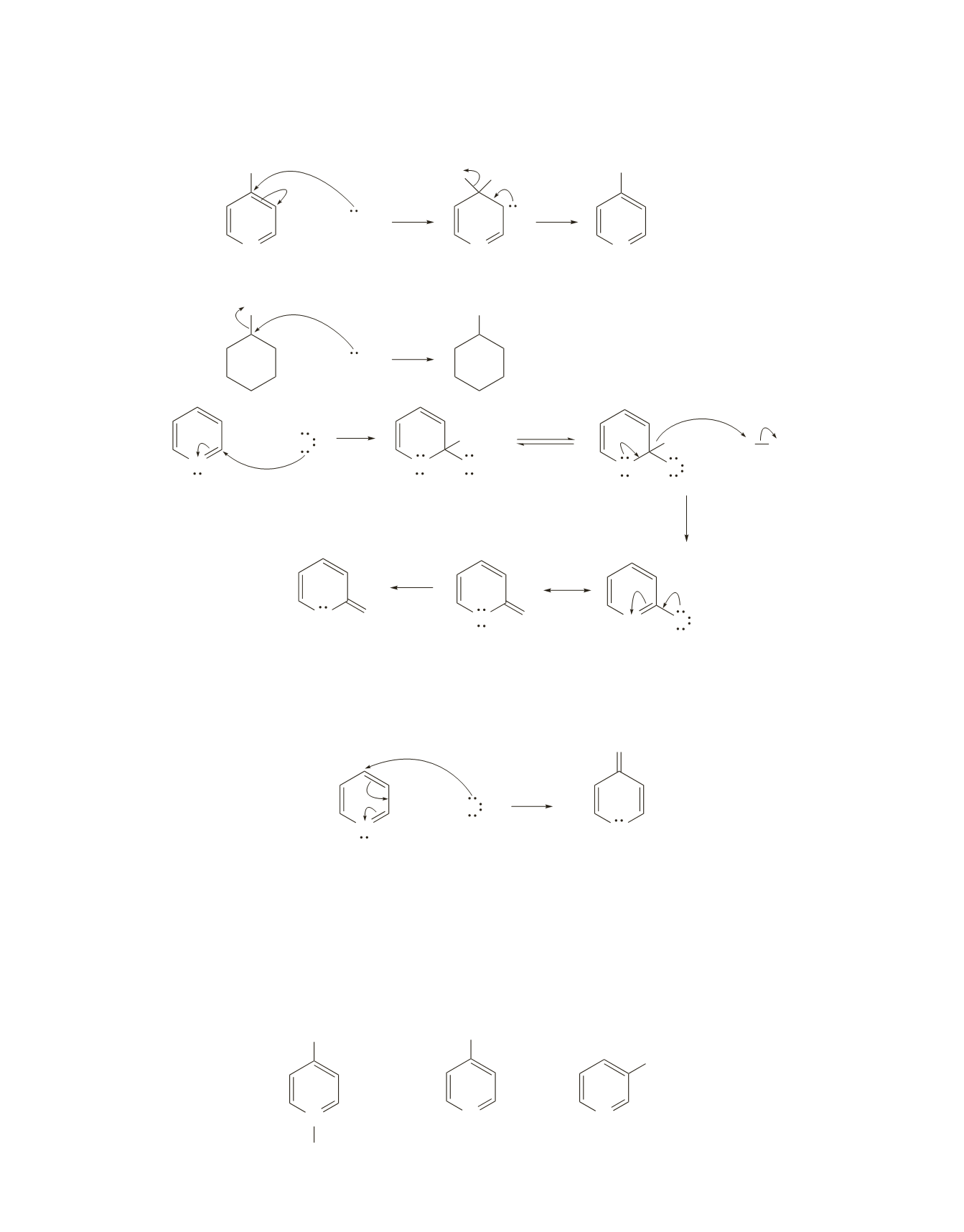
656 Chapter 19
Copyright © 2017 Pearson Education, Inc.
11.
The first reaction is a two-step nucleophilic aromatic substitution reaction
1
S
N
Ar
2
.
N
Cl
+
NH
2
N
Cl
NH
2
N
+
NH
2
Cl
_
_
_
Cl
+
NH
2
+
NH
2
_
Cl
_
N
N
Cl
+
NH
2
N
Cl
NH
2
N
+
NH
2
Cl
_
_
_
Cl
+
NH
2
+
NH
2
_
Cl
_
The second reaction is a one-step nucleophilic substitution reaction (S
N
2).
NH
H
2
_
_
12.
a.
N
+
N
H
OH
N
H
O
H OH
+
H
2
O
_
_
a.
N
H
O
N O
_
N O
_
+
+
+
H
2
O
2-pyridone
HO
_
_
HO
−
HO
−
HO H
2
−
The hydride ion
is a better leaving
group than O
2
−
.
b.
4-Pyridone is also formed because nucleophilic addition of hydroxide ion can take place at the
4-position as well as at the 2-position. It proceeds by the same mechanism as the one shown in part
a
for the formation of 2-pyridone.
N
+
HO
_
N
H
4-pyridone
O
H
2
O
13.
It is easiest to remove a proton from the methyl group of the
N
-alkylated pyridine because the electrons left
behind when the proton is removed can be delocalized onto the positively charged nitrogen. (A positively
charged nitrogen more readily accepts the delocalized electrons than does a neutral nitrogen.)
It is easier to remove a proton from 4-methylpyridine than from 3-methylpyridine because in the former,
the electrons left behind when the proton is removed can be delocalized onto the electronegative nitrogen.
In contrast, in 3-methylpyridine the electrons can be delocalized onto only carbons.
N
CH
2
CH
3
CH
3
N
CH
3
N
CH
3
>
>
+
I
−
















