
658 Chapter 19
Copyright © 2017 Pearson Education, Inc.
15.
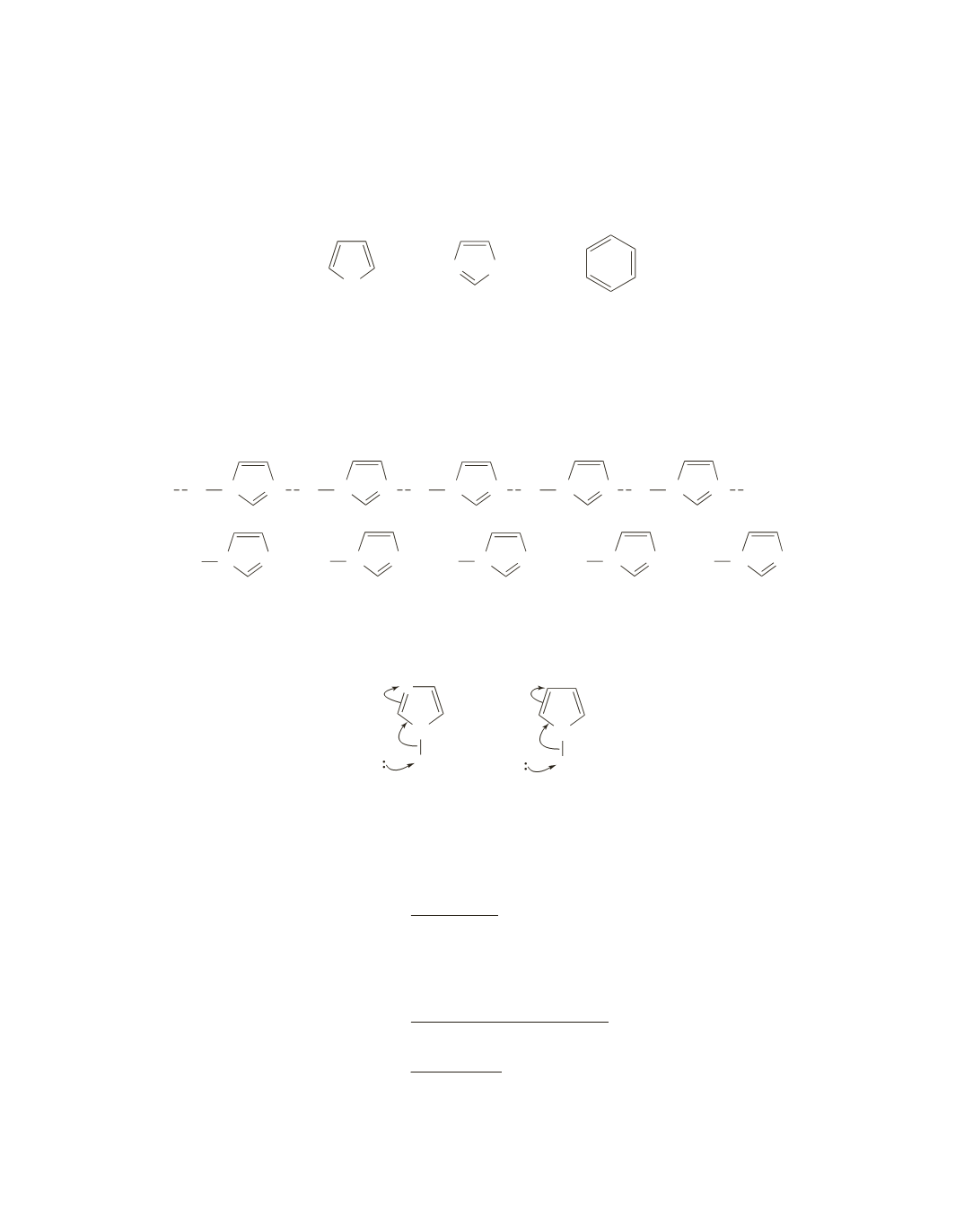
Pyrrole and imidazole are both more reactive than benzene because each reacts with an electrophile to form a
carbocation intermediate that is stabilized by resonance electron donation into the ring by a nitrogen atom.
Pyrrole is more reactive than imidazole because the second nitrogen atom of imidazole cannot donate elec-
trons into the ring by resonance but can withdraw electrons from the ring inductively.
N
H
pyrrole
imidazole
benzene
>
NH N
>
16.
Imidazole forms intermolecular hydrogen bonds, whereas
N
-methylimidazole cannot form hydrogen bonds
because it does not have a hydrogen bonded to a nitrogen.
Because the hydrogen bonds have to be broken in order for the compound to boil, imidazole has a higher
boiling point.
N N
N N
N
N N
H
N
N CH
3
H
H
H
H
N
N
N
N N
N
N N
N
CH
3
CH
3
CH
3
CH
3
N
N
17.
The second nitrogen in imidazole, onto which the electrons left behind when the proton is removed can be
delocalized, causes imidazole to be a stronger acid than pyrrole because nitrogen is more electronegative
than carbon.
N
imidazole
pyrrole
p
K
a
=
14.4
p
K
a
=
17
N
H B
N
H B
18.
For the derivation of the equation used in this problem, see Special Topic I in this
Study Guide and
Solutions Manual
.
fraction of imidazole in the acidic form
=
3
H
+
4
K
a
+
3
H
+
4
pH
=
7.4;
3
H
+
4
=
4.0
*
10
-
8
p
K
a
=
6.8;
K
a
=
1.58
*
10
-
7
1
see Table 20.1
2
fraction of imidazole in the acidic form
=
4.0
*
10
-
8
1.58
*
10
-
7
+
4.0
*
10
-
8
=
4.0
*
10
-
8
1.98
*
10
-
7
=
0.20
percent of imidazole in the acidic form
=
20
,
















