
512 Chapter 15
Copyright © 2017 Pearson Education, Inc.
91.
N
C
O
NO
2
O
NO
2
HO
N
C
OH
O
+
+
+
N
H
O
O
HO
P
O
or NH
NO
2
O
−
O
H
2
O
a.
c.
b.
HO
O
HO
O
C
O
HO O
NO
2
δ
−
δ
−
C
O
HO O
NO
2
δ
−
δ
−
transition state if formation of the
tetrahedral intermediate is rate-limiting
transition state if collapse of the
tetrahedral intermediate is rate-limiting
92.
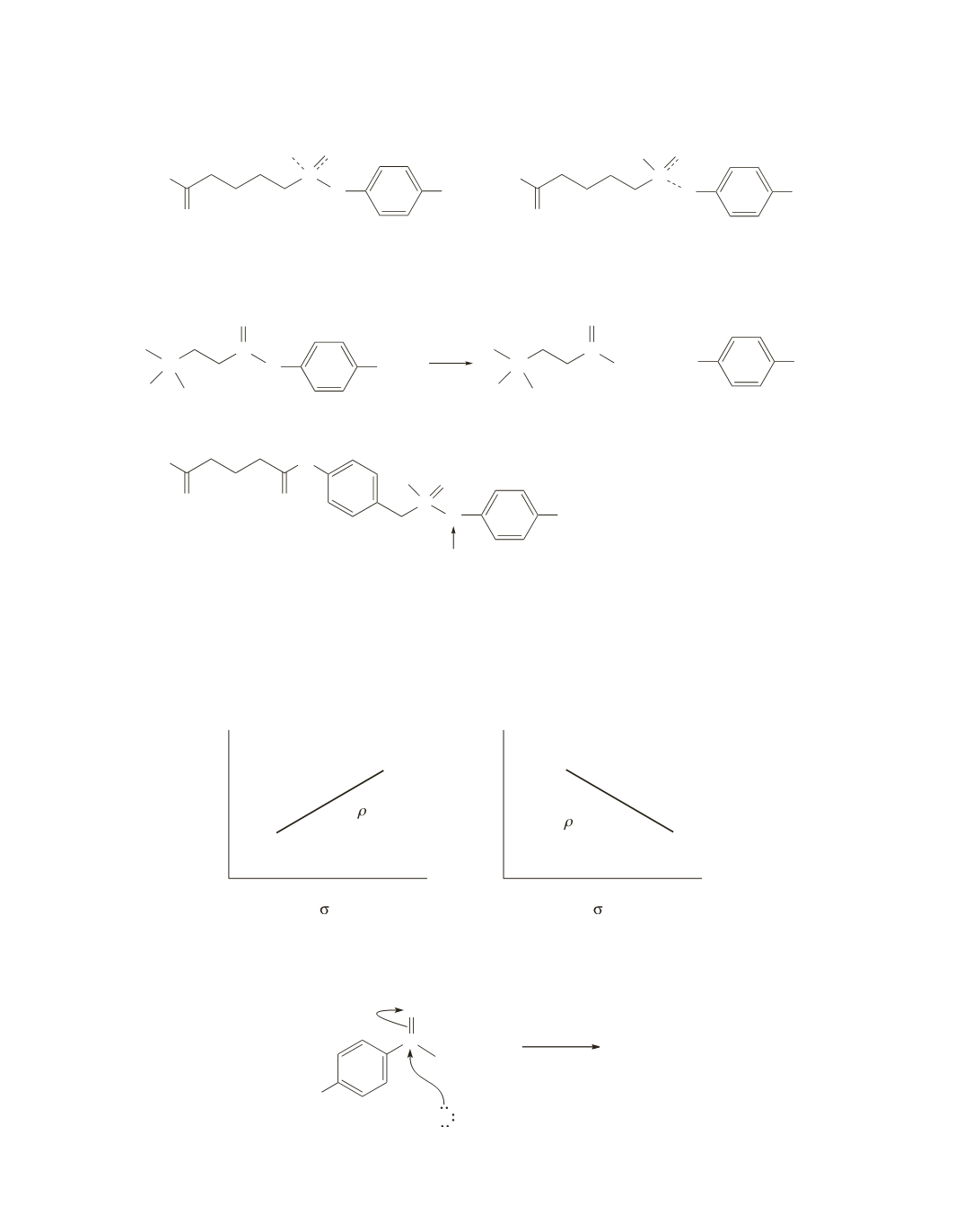
Because electron-withdrawing substituents have positive substituent constants and electron-donating
substituents have negative substituent constants, a reaction with a positive
r
value is one in which
compounds with electron-withdrawing substituents react more rapidly than compounds with electron-
donating substituents, and a reaction with a negative
r
value is one in which compounds with electron-
donating substituents react more rapidly than compounds with electron-withdrawing substituents.
electron withdrawal increases
the rate of the reaction
−
2
0
2
−
2
0
2
= +
= −
electron donation increases
the rate of the reaction
log rate
log rate
a.
In the hydroxide-ion-promoted hydrolysis of a series of ethyl benzoates, electron-withdrawing substituents
increase the rate of the reaction by increasing the amount of positive charge on the carbonyl carbon, thereby
making it more readily attacked by hydroxide ion. The
r
value for this reaction is, therefore, positive.
C
O
OCH
2
CH
3
Y
_
HO
















