
508 Chapter 15
Copyright © 2017 Pearson Education, Inc.
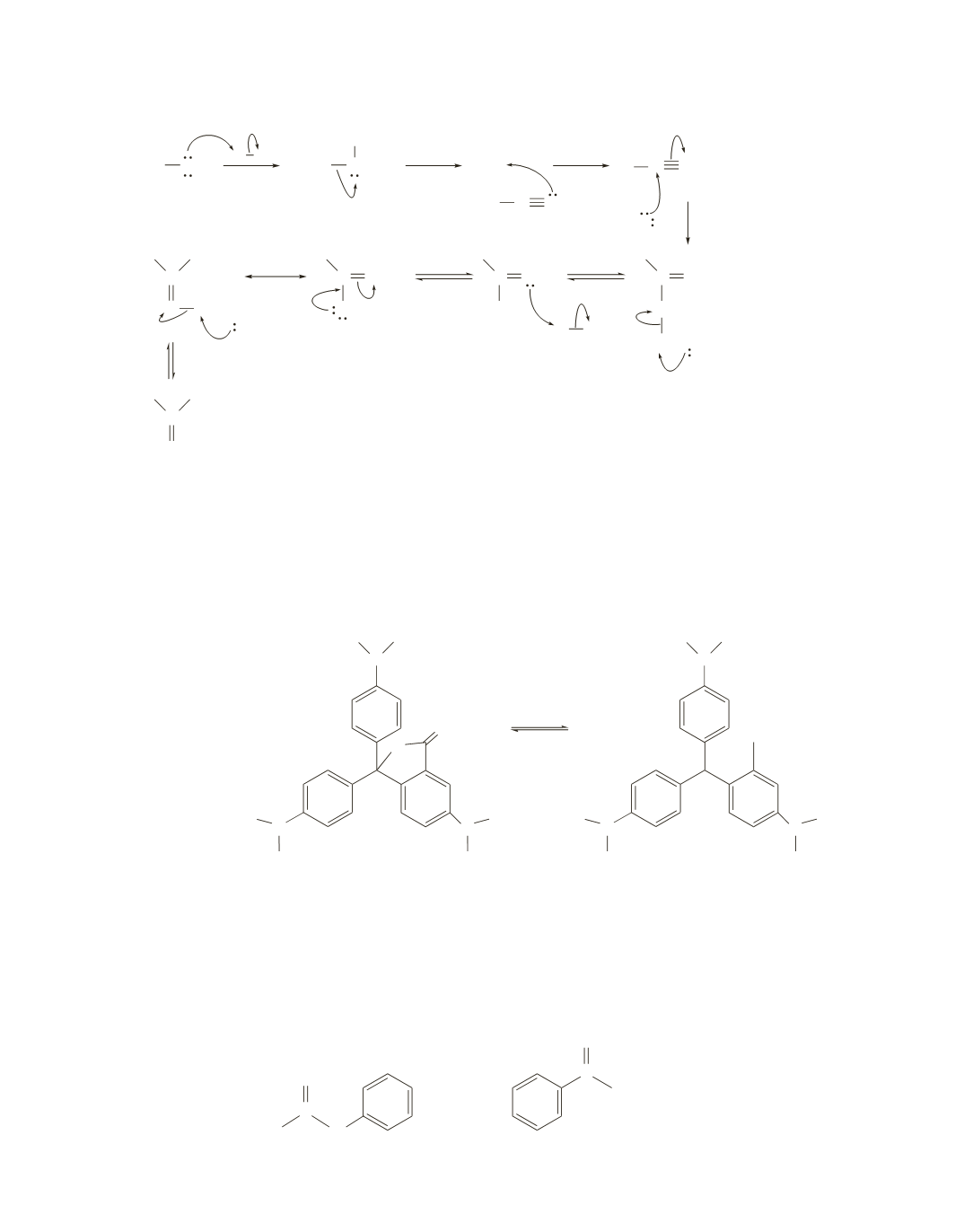
82.
a.
R OH
H B
+
R OH
H
+
+
+
R
R NR
CR N
C
H
2
O
O H
+
C NHR
OH
NR
OH
NR
OH
+
+
H
H B
+
B
C
R NHR
O
HB
+
+
+
B
C
R NHR
R
C
R
C
R
b.
The Ritter reaction does not work with primary alcohols because primary alcohols do not form
carbocations.
c.
The only difference in the two reactions is the electrophile that attaches to the nitrogen of the nitrile: it
is a carbocation in the Ritter reaction and a proton in the acid-catalyzed hydrolysis of a nitrile.
83.
Before the bond breaks, the three rings are not conjuated. The rings are conjugated in the carbocation.
Therefore, the carbocation is a highly colored species.
N
O
O
N
N
COOH
N
N
N
H
+
HO
−
+
84.
Each of the NMR spectra has signals between about 7 and 8 ppm (integrating to 5H), indicating that the
compound has a benzene ring with one substituent. There is one additional signal that is a singlet. From
the molecular formulas, it can be determined that the esters have the following structures. The singlet in
each spectrum is due to a methyl group. Because the methyl group is at a higher frequency in the lower
spectrum, it is the spectrum of the compound on the right, since its methyl group is adjacent to an electron-
withdrawing oxygen. The compound on the left will be hydrolyzed more rapidly because its OR group is a
weaker base. (The p
K
a
of phenol is
∼
10; the p
K
a
of methanol is
∼
16.)
OCH
3
O
CH
3
C
O
C
O
















