
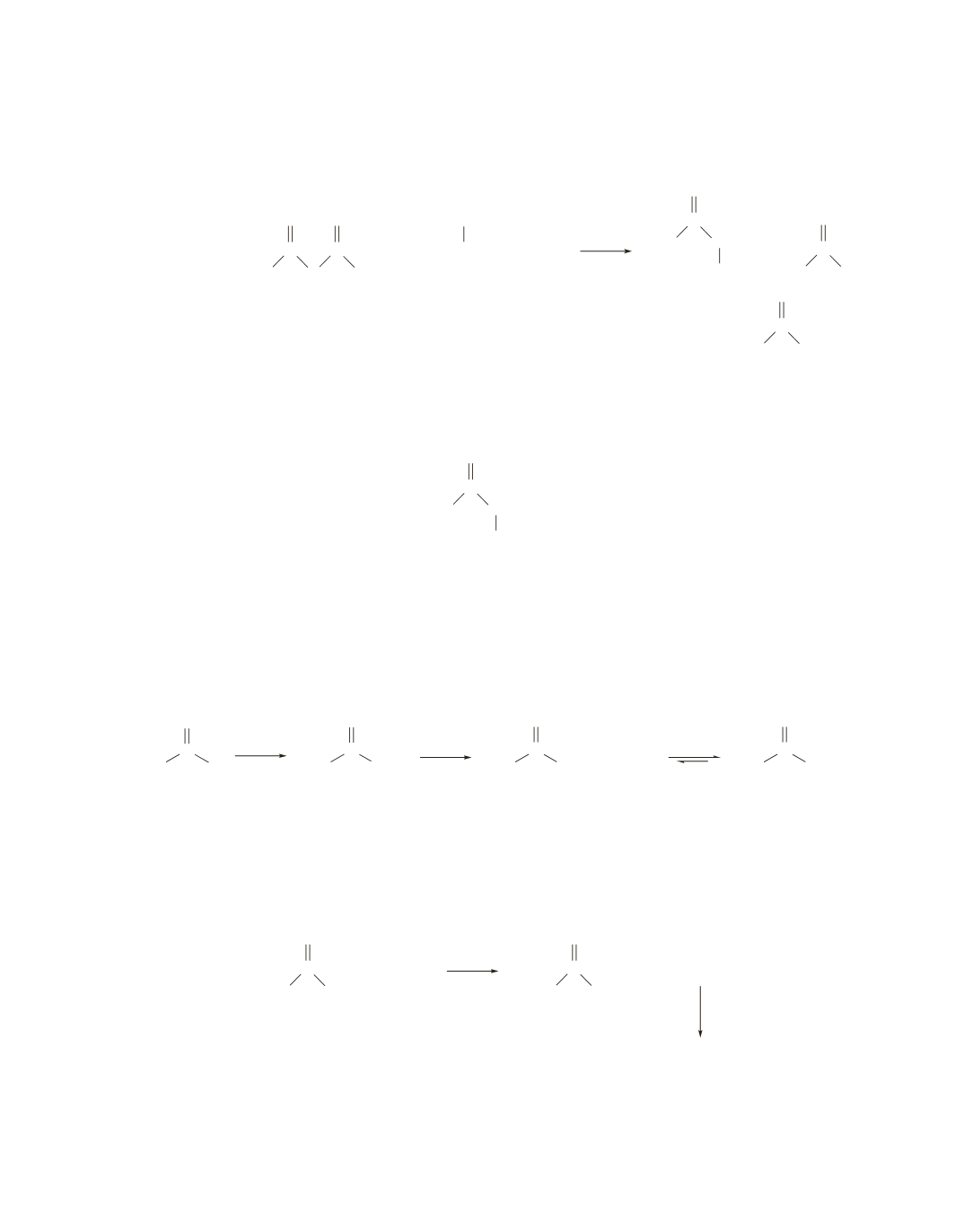
Chapter 15 505
Copyright © 2017 Pearson Education, Inc.
74.
a.
The amino alcohol has two groups that can be acetylated by acetic anhydride, the NH
2
group and the
OH group.
+
–
CH
3
O
CH
3
O CH
3
+
CH
3
CHCH
2
CH
2
OH
NH
2
CH
3
CHCH
2
CH
2
O
NH
CH
3
CH
3
C 2
2
O
C
O
C
O
C
O
C
O
b.
Because the NH
2
group is a better nucleophile than the OH group, it will be the first group that is
acetylated. Therefore, if the reaction is stopped before it is half over, the product will have only its NH
2
group acetylated.
CH
3
CHCH
2
CH
2
OH
NH
CH
3
C
O
75.
If the amine is tertiary, the nitrogen in the amide product cannot get rid of its positive charge by losing a
proton. An amide with a positively charged nitrogen is very reactive toward nucleophilic acyl substitution
because the positively charged nitrogen-containing group is an excellent leaving group. Therefore, if water
is added, it will immediately react with the amide and, because the
+
NR
3
group is a better leaving group
than the OH group, the
+
NR
3
group will be eliminated. The product will be a carboxylic acid, which will
lose its acidic proton to the amine.
C
O
−
CH
3
+
+
HNR
3
O
O
C
Cl
CH
3
2 NR
3
C
NR
3
CH
3
+
H
2
O
C
OH
CH
3
+
NR
3
+
HNR
3
Cl
−
O
O
76.
The reaction of methylamine with propionyl chloride generates a proton that will protonate unreacted
amine, thereby destroying the amine’s nucleophilicity. If two equivalents of CH
3
NH
2
are used, one
equivalent will remain unprotonated and be able to react as a nucleophile with propionyl chloride to form
N-
methylpropanamide.
CH
3
CH
2
+
CH
3
NH
2
+
H
+
+
Cl
−
CH
3
NH
2
CH
3
NH
3
+
C
Cl
O
CH
3
CH
2
NHCH
3
C
O
















