
500 Chapter 15
Copyright © 2017 Pearson Education, Inc.
60.
The offset in the NMR spectrum shows that there is a broad signal at
∼
10 ppm, which is characteristic of a
COOH group. The two triplets and the multiplet are characteristic of a propyl group.
CH
3
CH
2
CH
2
t
m t
Therefore, the product of the hydrolysis reaction is butanoic acid.
O
C
CH
3
CH
2
CH
2
OCH
3
reactant
OH
CH
3
CH
2
CH
2
C
O
hydrolysis product
The molecular formula shows that the reactant has one more carbon than butanoic acid; the IR spectrum
shows that it is an ester. Because butanoic acid is formed from acid hydrolysis of the ester, the reactant
must be the methyl ester of butanoic acid.
61.
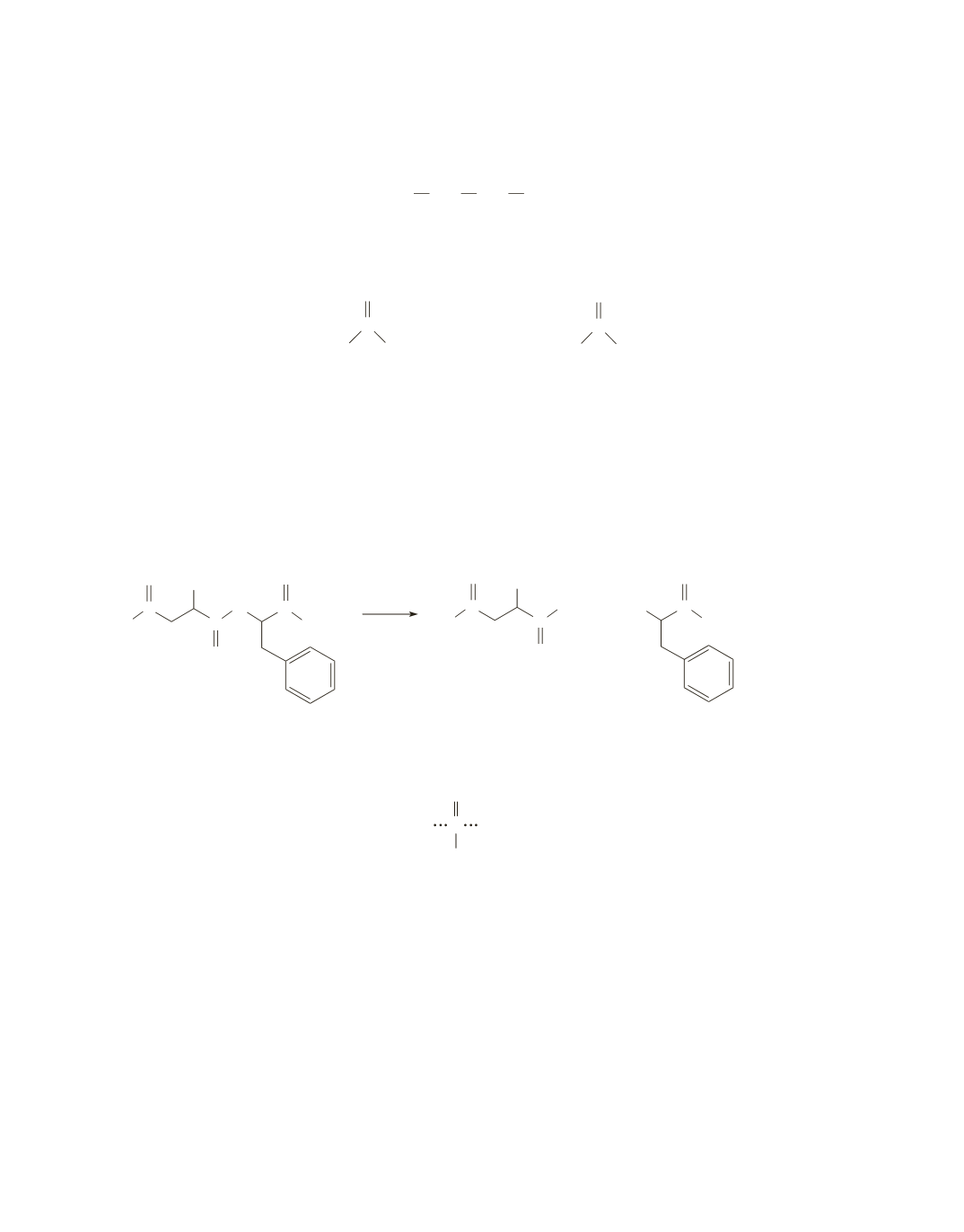
Aspartame has an amide group and an ester group that will be hydrolyzed in an aqueous solution of HCl.
Because the hydrolysis is carried out in an acidic solution, the carboxylic acid groups and the amino groups
in the products will be in their acidic forms.
_
+
+
O
C
O
C
N C
OCH
3
NH
3
O
O
H
+
HO
C
O
C
NH
3
O
OH
OH OH
+
HCl
H
2
O
C
CH
3
O
H
3
N
+
62.
If it were an S
N
2 reaction, the ester would be the only species that would be isotopically labeled and there
would be no unlabeled ester because the carbonyl group would not have participated in the reaction.
HO OR C
R
O
–
–
















