
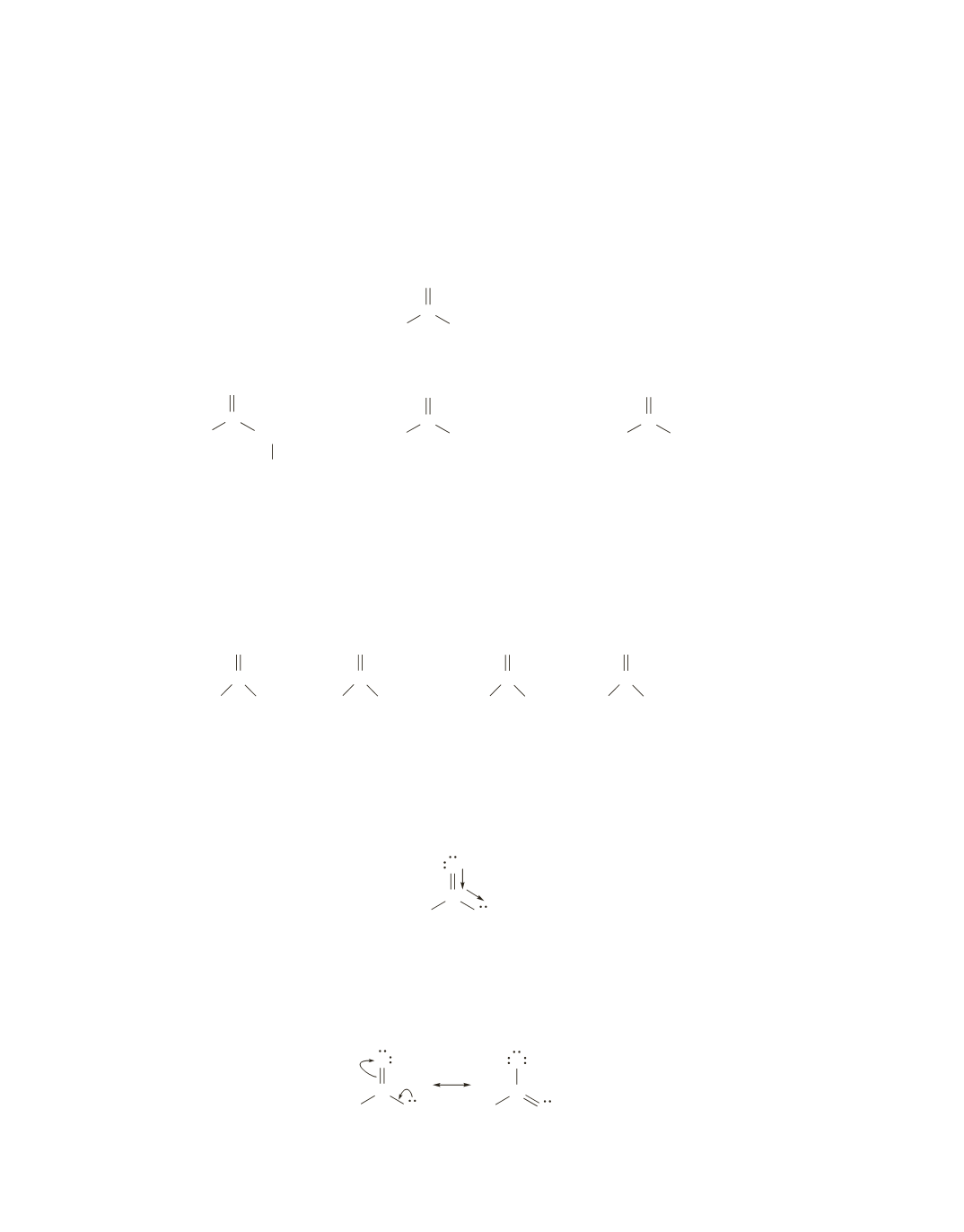
498 Chapter 15
Copyright © 2017 Pearson Education, Inc.
55.
Propyl formate is easy to distinguish because it is the only ester that shows four signals. The other three
esters each show three signals.
Isopropyl formate can be distinguished by its unique splitting pattern: a singlet, a doublet, and a septet.
The splitting patterns of the other two esters are the same: a singlet, a triplet, and a quartet. They can be
distinguished because the highest frequency signal is due to the protons attached to the carbon that is next
to the oxygen. For ethyl acetate, it is a quartet, whereas for methyl propionate, it is a singlet.
H OCH
2
CH
2
CH
3
4 signals
H OCHCH
3
CH
3
OCH
2
CH
3
CH
3
CH
2
OCH
3
CH
3
C
O
C
O
C
O
C
O
3 signals
3 signals
3 signals
singlet, doublet, septet
singlet, triplet, quartet singlet, triplet, quartet
The highest frequency The highest frequency
signal is a quartet.
signal is a singlet.
56.
The carbonyl IR absorption band decreases in the order:
>
>
C
OCH
CH
3
3
O
>
C
Cl
CH
3
O
C
CH
3
H
O
C
NH
CH
3
2
O
The carbonyl IR absorption band of the acyl chloride occurs at the highest frequency because there is
essentially no electron delocalization from a lone pair on chlorine and the chlorine withdraws electrons
inductively. The carbonyl IR absorption band of the ester is next because the predominant effect of the
oxygen of an ester is inductive electron withdrawal. Therefore, it takes more energy to stretch the bond.
(See page 592 of the text.)
O
C
Y R
The carbonyl group of the amide stretches at the lowest frequency because the nitrogen is less
electronegative than oxygen, so it is better able to accommodate a positive charge. Therefore, the resonance
contributor on the right makes a significant contribution to the overall structure. The larger the contribution
from the resonance contributor on the right, the greater the single bond character of the C
“
O group.
O
C
Y R
O
C
Y R
−
+
















