
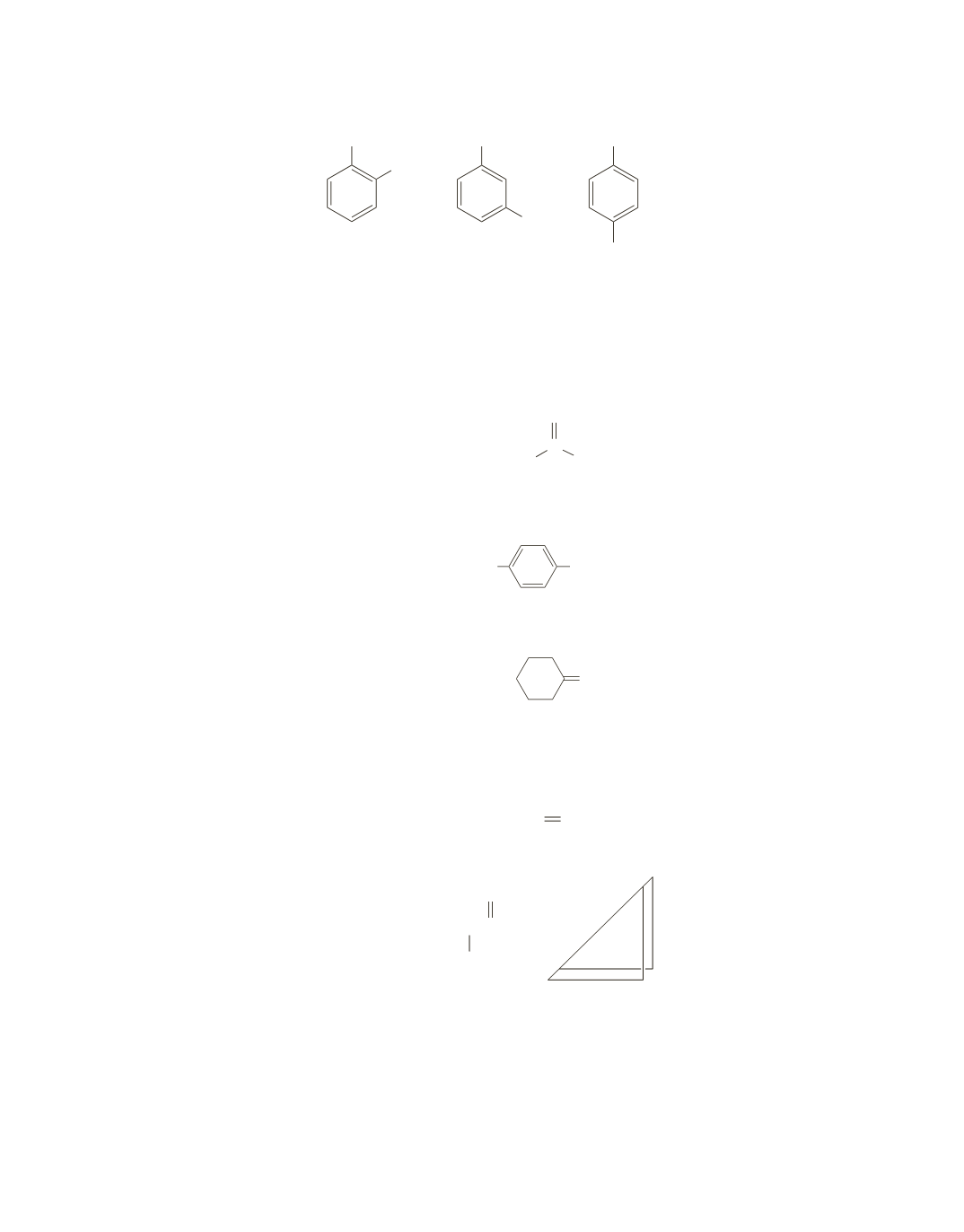
Chapter 14 469
Copyright © 2017 Pearson Education, Inc.
43.
NO
2
NO
2
NO
2
NO
2
NO
2
NO
2
a.
1
H
NMR
two signals
three signals
one signal
b.
13
C
NMR three signals
four signals
two signals
44.
a.
The signal at 210 ppm is for the carbonyl carbon of a ketone. There are 10 other carbons in the com-
pound but only 5 other signals. This suggests that the compound is a symmetrical ketone with identical
five-carbon alkyl groups.
O
C
CH
3
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
3
b.
Because there are only four signals for the six carbons of the benzene ring (the signals between 110
and 117 ppm), the compound must be a 1,4-disubstituted benzene with two different substituents.
CH
2
CH
3
Br
c.
The signal at 212 ppm is for the carbonyl carbon of a ketone. There are five other carbons in the com-
pound but only three other signals. This suggests that the compound is a symmetrical cyclic ketone.
O
d.
The molecular formula indicates that the compound has one double bond or one ring. The presence of a
signal at 130 ppm indicates the presence of
sp
2
carbons; thus, the compound must have a double bond.
Each of the two
sp
2
carbons must be bonded to the same groups because the six carbons exhibit only three
signals. Whether the compound is
cis
-3-hexene or
trans
-3-hexene cannot be determined from the spectrum.
CH
3
CH
3
CH
2
CH
2
CH CH
45.
If the triangles shown below are drawn on the spectrum, you will be able to identify the coupled protons.
CH
3
CHCCH
2
CH
3
CH
3
O
b d c a
b
B
A
Point
A
shows that the
a
protons are split by the
c
protons.
Point
B
shows that the
b
protons are split by the
d
protons.
46.
Cross peak X tells you that the hydrogens that produce the signal at
1.0
ppm in the
1
H
NMR spectrum are
bonded to the carbon that produces the signal at
19
ppm in the
13
C
NMR spectrum.
















