
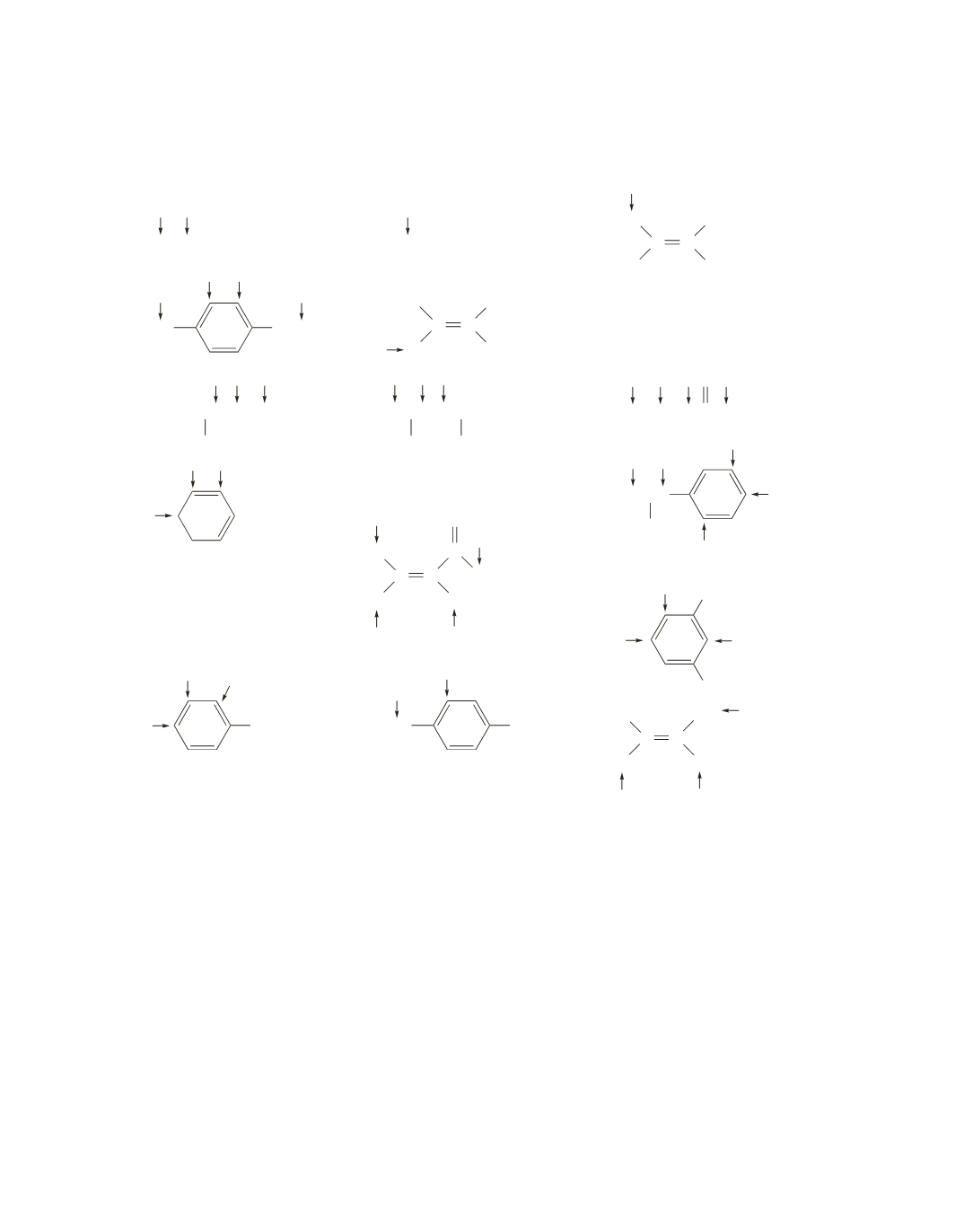
466 Chapter 14
Copyright © 2017 Pearson Education, Inc.
27.
s
=
singlet, d
=
doublet, t
=
triplet, q
=
quartet, quin
=
quintet, d of d
=
doublet of doublets, and
m
=
multiplet.
CH
3
CH
2
CH
2
CH
3
t q
a.
BrCH
2
CH
2
Br
s
b.
CH
3
OCH
3
d d
s
s
d.
d
t
Br
Br
s
l.
CH
3
CH
3
s
s
n.
NO
2
d of d
t
d
m.
1 2
3
4 5
6
m d
d
j.
C C
H
Cl
H
Cl
s
e.
CH
3
CH
2
CH
2
CCH
3
Ot
t m s
f.
CH
3
CH
Br
q d
d
t
d of d
i.
g.
CH
3
CH
2
CHCH
2
CH
3
Cl
m t
quin
CH
3
CHCH
2
CHCH
3
CH
3
CH
3
m t
d
h.
C C
H
H
Cl
Cl
s
c.
(Remember that equivalent
hydrogens do not split
each other’s signal.)
The H on C-2 will not be split
by the H on C-3 because they
are equivalent. The Hs on C-6
will not be split by the Hs on
C-5 because the Hs on C-5
and C-6 are equivalent.
28.
Each spectrum is described going from left to right.
a.
BrCH
2
CH
2
CH
2
CH
2
Br
triplet triplet (This triplet will not be split by the adjacent equivalent protons.)
b.
two triplets (close to each other) singlet multiplet
(The table “
1
H
NMR Chemical Shifts” in Appendix V indicates that a methylene group adjacent to an
RO and a methylene group adjacent to a Br appear at about the same place.)
c.
singlet (Equivalent Hs do not split each other’s signals.)
d.
quartet singlet triplet
e.
three singlets
f.
three doublets of doublets
g.
quartet triplet
h.
singlet quartet triplet
i.
doublet multiplet doublet
j.
triplet quintet
k.
doublet doublet of septets doublet
l.
triplet singlet quintet
m.
singlet
n.
singlet (Equivalent Hs do not split each other’s
signals.)
o.
singlet
p.
quintet, multiplet, multiplet
C
O
CC
H
d
H
H
d
m
H
d
CH d
CC
C1
3
H
d
H
d
k.
o.
















