
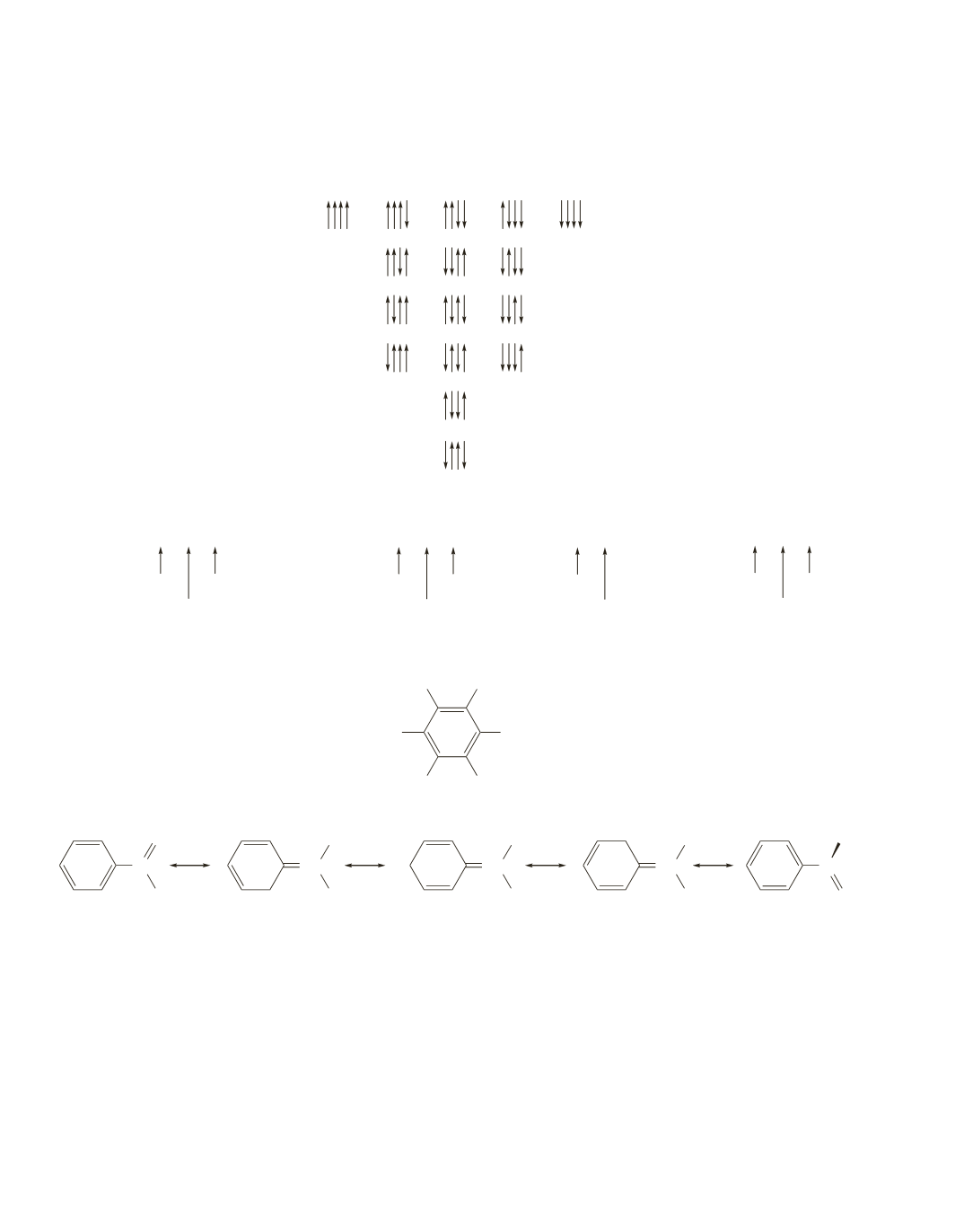
464 Chapter 14
Copyright © 2017 Pearson Education, Inc.
b.
A quintet is caused by coupling to four equivalent protons on adjacent carbons. The four protons can
be aligned in five different ways. The following possible arrangements for the alignment of four pro-
tons explain why the relative intensities of a quintet are 1:4:6:4:1.
23.
a.
three signals
triplet triplet
multiplet
CH
3
CH
2
CH
2
CH
2
CH
2
CH
3
b.
three signals
ICH
2
CH
2
CH
2
Br
triplet triplet
multiplet
c.
two signals
ClCH
2
CH
2
CH
2
Cl
triplet
quintet
d.
three signals
ICH
2
CH
2
CHBr
2
triplet triplet
multiplet
24.
The nitro group withdraws electrons inductively and by resonance.
H
H H
H H
a
c
b
NO
2
N
O
O
−
O
−
O
−
+
+
+
N
+
+
N
+
N
+
O
−
O
−
O
−
O
−
N
O
O
−
The
a
protons are at the lowest frequency because the nitro group withdraws electrons only inductively
from the carbons to which they are attached.
The
b
and
c
protons are at a higher frequency because the nitro group withdraws electrons both inductively
and by resonance from the carbons to which they are attached.
The
c
protons are at the highest frequency because the nitro group withdraws electrons inductively more strongly
from the carbons to which the
c
protons are attached since those carbons are closest to the nitro group.
















