
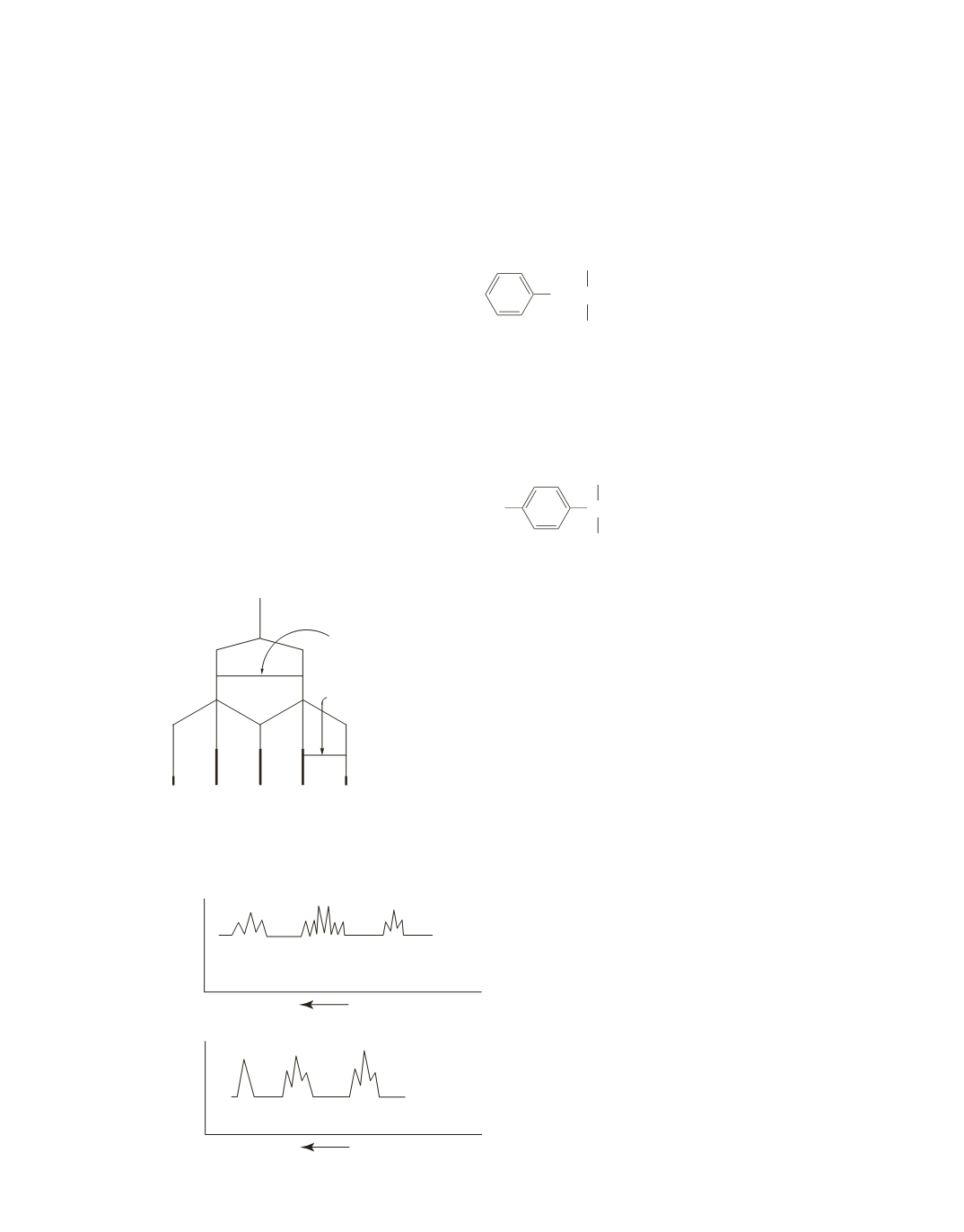
Chapter 14 475
Copyright © 2017 Pearson Education, Inc.
67.
The signals at
7.2
ppm indicate that the compounds whose spectra are shown in parts
a
and
b
contain a
benzene ring. From the molecular formula, you know that each compound has five additional carbons.
a.
The integration of the benzene ring protons indicate that the ring is monosubstituted. The hydrogens
bonded to the five additional carbons in part
a
must all be accounted for by two singlets with integral
ratios of
2:9,
indicating that the compound is
2,2-dimethyl-1-phenylpropane
.
CH
2
CCH
3
CH
3
CH
3
2,2-dimethyl-1-phenylpropane
b.
The integration of the benzene ring protons indicate that the ring is 1,4-disubstituted. The two singlets
in the spectrum in part
b
have integral ratios of ~1:3, indicating a methyl substituent and a
tert
-butyl
substituent. Therefore, the compound is
1-
tert
-butyl-4-methylbenzene
.
CCH
3
CH
3
CH
3
CH
3
1-
tert
-butyl-4-methylbenzene
68.
ve signals
J
ba
J
bc
69.
Oxygen is more electronegative than chlorine. (See Table 1.3 on page 10 of the text.)
a.
c
a
b
c a b
ClCH
2
CH
2
OH
triplet
triplet
frequency
doublet
of
triplets
b.
frequency
singlet
c
a
b
triplet
triplet
















