
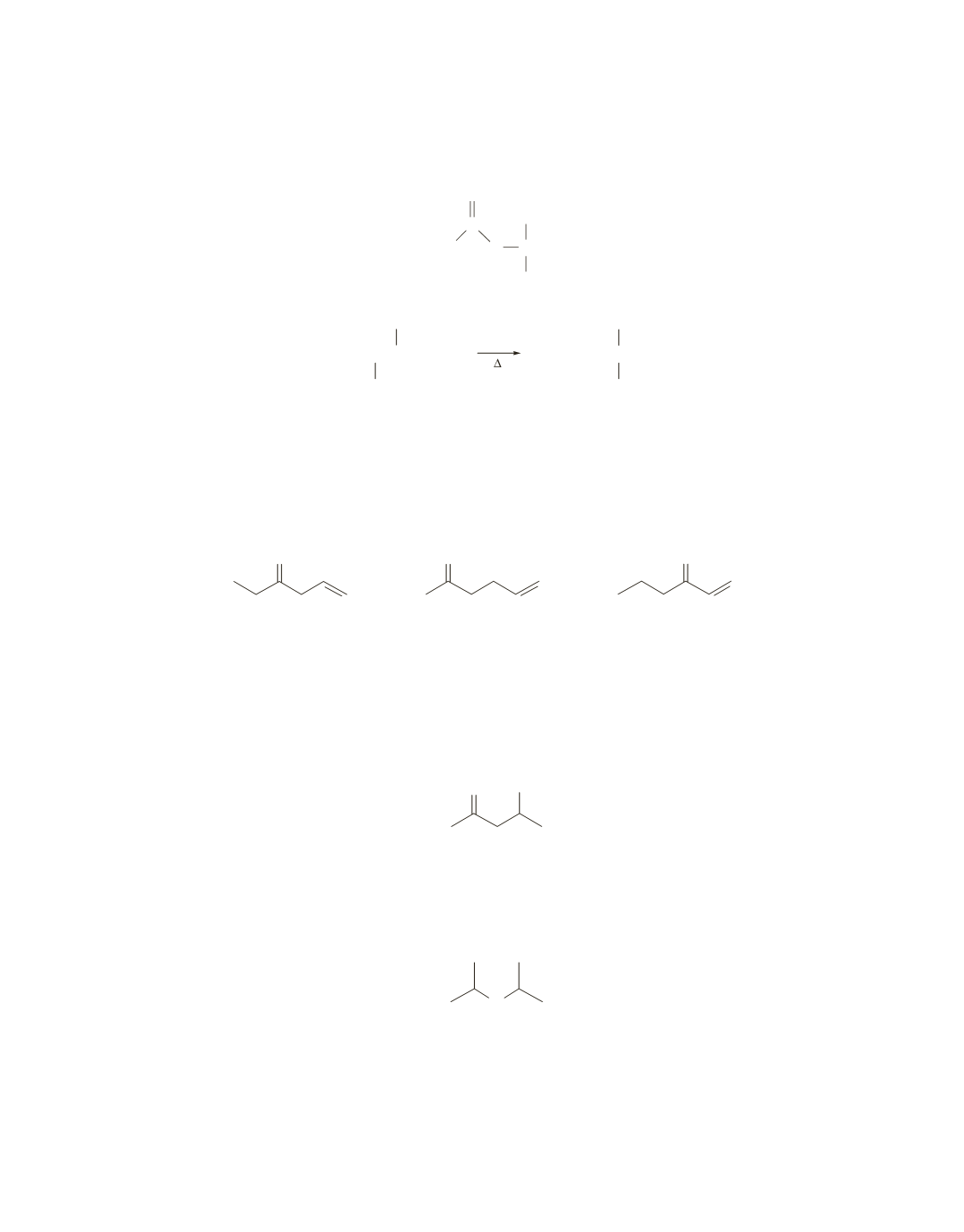
478 Chapter 14
Copyright © 2017 Pearson Education, Inc.
d.
The group attached to the oxygen in the fourth spectrum has only one kind of hydrogen. The carbon
attached to the oxygen is not bonded to any hydrogens, because there is no signal at
4.0.
C
O
O
CCH
3
CH
3
CH
3
CH
3
75.
CH
3
CHCHCH
3
OH
CH
3
HBr
CH
3
CH
2
CCH
3
CH
3
Br
compound
A
compound
B
76.
The DEPT
13
C
NMR spectrum indicates that the compound has six carbons. The molecular formula shows
that the compound has six carbons and an oxygen. The signal at 220 ppm suggests a ketone. The following
ketones have the single
CH
3
group, the single CH group, and the three
CH
2
groups indicated by the DEPT
spectrum. The highest frequency signals are a
CH
2
group and a CH group, indicating that these are the
groups attached to the carbonyl carbon. Thus, the compound on the right is responsible for the spectrum.
O
O
or
O
or
77.
a.
The IR spectrum indicates that the compound is a ketone. The molecular formula shows that there are
12 hydrogens in the compound. Therefore, the signals in the NMR spectrum are due to 2, 4, and 6 protons.
The doublet in the NMR spectrum at
0.9
ppm that integrates to 6 protons suggests an isopropyl group.
There is a singlet at
2.1
ppm on top of a multiplet, which is due to the methine proton of an isopropyl
group plus a three-hydrogen singlet for a methyl group adjacent to the carbonyl carbon. The doublet at
2.2
ppm that integrates to 2 protons suggests a
CH
2
group adjacent to the carbonyl group that couples to
an adjacent methine proton. Knowing that the compound contains six carbons helps in the identification.
O
b.
The IR spectrum indicates that this oxygen-containing compound is not a carbonyl compound or an
alcohol; the absorption band at
1000 cm
-
1
suggests that it is an ether. The doublet at
1.1
ppm in
the
1
H
NMR spectrum that integrates to 6 protons and the septet that integrates to 1 proton indicate an
isopropyl group. Because there are no other signals in the NMR spectrum, the compound must be a
symmetrical ether. The compound is
diisopropyl ether
.
O
















