
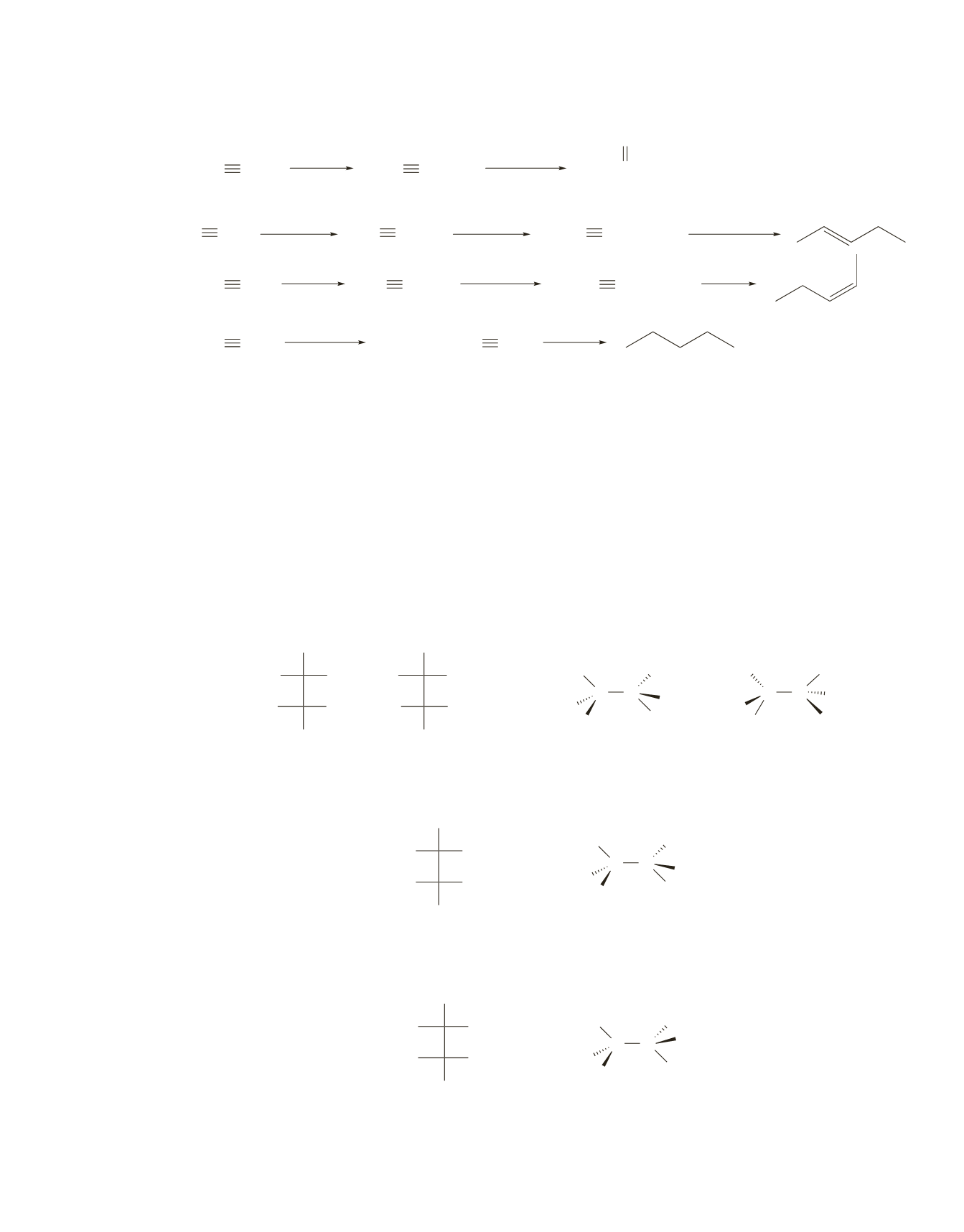
Chapter 7 253
Copyright © 2017 Pearson Education, Inc.
c.
HC CH
1. NaNH
2
2. CH
3
Br
HC CCH
3
H
2
O, H
2
SO
4
HgSO
4
CH
3
CCH
3
O
d.
HC CH
1. NaNH
2
2. CH
3
Br
HC CCH
3
CH
3
C CCH
2
CH
3
1. NaNH
2
2. CH
3
CH
2
Br
Na
NH
3
(liq), 78
°
C
−
e.
HC CH
1. NaNH
2
2. CH
3
Br
HC CCH
3
1. NaNH
2
2. CH
3
CH
2
Br
CH
3
C CCH
2
CH
3
H
2
Lindlar
catalyst
f.
HC CH
1. NaNH
2
2. CH
3
CH
2
CH
2
Br
excess H
2
Pd/C
CH
3
CH
2
CH
2
C CH
47.
The first equilibrium lies to the right because HOOH
1
p
K
a
=
11.6
2
is a stronger acid than H
2
O
1
p
K
a
=
15.7
2
.
The second equilibrium lies to the left because the alkyne
1
p
K
a
=
25
2
is a weaker acid than HOOH
1
p
K
a
=
11.6
2
.
48.
a.
(
Z
)-3,6-dimethyl-2-hepten-4-yne
b.
5-
tert
-butyl-2-methyl-3-octyne
c.
4,4-dimethyl-1-nonen-6-yne-3-ol
d.
4-(2-methylbutyl)-7-heptyne-3-amine
49.
a.
Syn addition of H
2
forms
cis
-2-butene; when Br
2
adds to
cis
-2-butene, the threo pair of enantiomers is
formed.
CH
3
H Br
CH
3
Br
H
CH
3
Br
H
CH
3
H Br
+
or
CH
3
CH
3
H
H
Br
Br
C C
H
Br
CH
3
Br
CH
3
H C C
b.
Reaction with sodium and liquid ammonia forms
trans-
2-butene; when Br
2
adds to
trans
-2-butene, a
meso compound is formed.
CH
3
H Br
CH
3
H Br
or
CH
3
Br
H
H
Br
CH
3
C C
c.
Anti addition of Cl
2
forms
trans
-2,3-dichloro-2-butene; when Br
2
adds to
trans-
2,3-dichloro-2-butene,
a meso compound is formed.
CH
3
Cl
Br
CH
3
Cl
Br
or
CH
3
Br
Cl
Cl
Br
CH
3
C C
















