
258 Chapter 7
Copyright © 2017 Pearson Education, Inc.
Chapter 7 Practice Test
1.
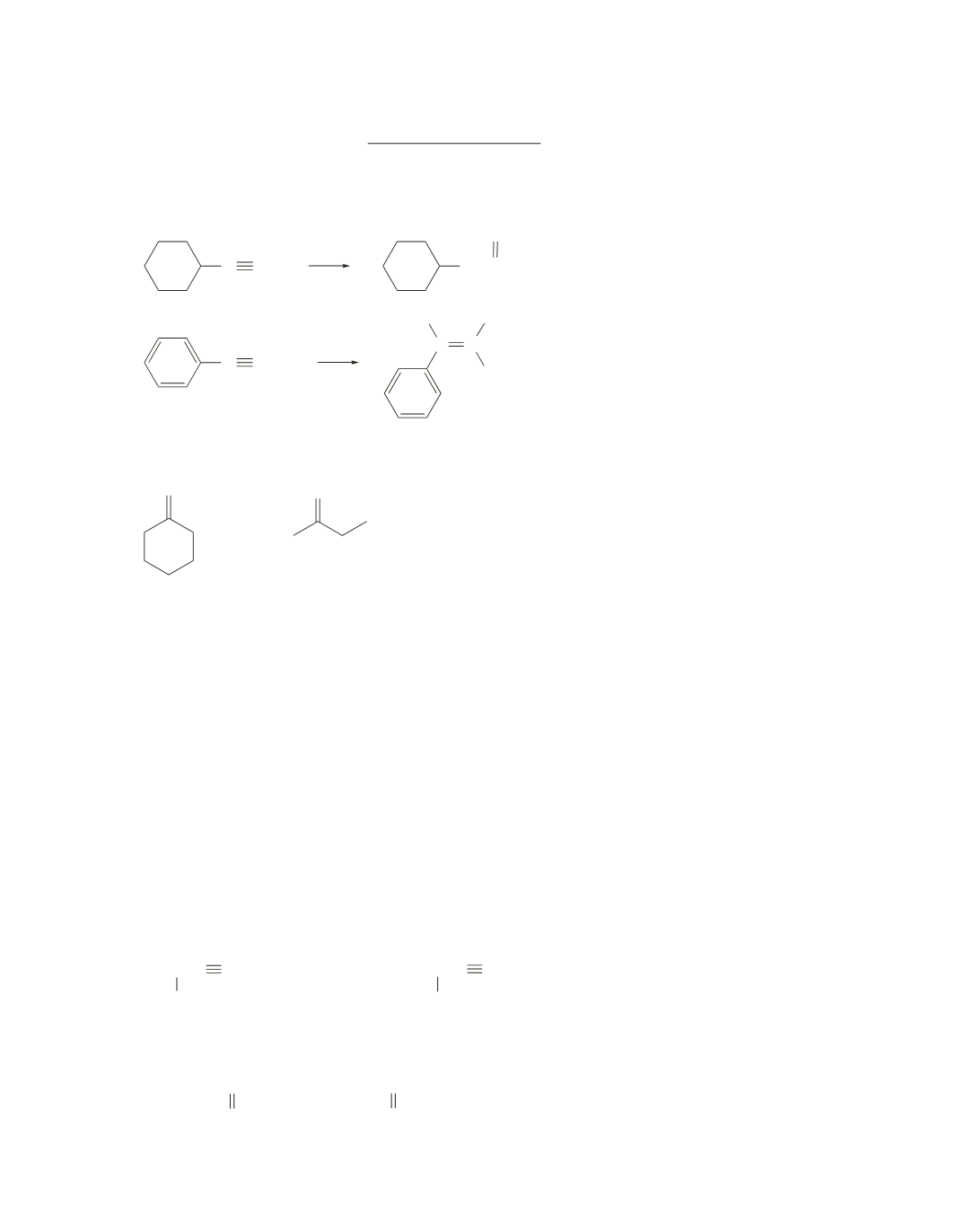
What reagents can be used to convert the given starting material into the desired product?
a.
C CH
CH
2
CH
O
b.
C CCH
3
C C
CH
3
H H
2.
Draw the enol tautomer(s) of the following ketones:
a.
O
b.
O
3.
Draw the structure for each of the following:
a.
sec
-butylisobutylacetylene
b.
2-methyl-1,3-cyclohexadiene
4.
Indicate whether each of the following statements is true or false:
a.
A terminal alkyne is more stable than an internal alkyne.
T
F
b.
Propyne is more reactive than propene toward reaction with HBr.
T
F
c.
1-Butyne is more acidic than 1-butene.
T
F
d.
An
sp
2
carbon is more electronegative than an
sp
3
carbon.
T
F
e.
The reactions of internal alkynes are more regioselective than the reactions of
terminal alkynes.
T
F
f.
Alkenes are more reactive than alkynes.
T
F
5.
What is each compound’s systematic name?
a.
CH
3
CHC CCH
2
CH
2
Br
CH
3
b.
CH
3
CHC CCH
2
CH
2
OH
CH
3
6.
What alkyne is the best reagent to use for the synthesis of each of the following ketones?
a.
CH
3
CH
2
CH
2
CCH
3
O
b.
CH
3
CH
2
CCH
2
CH
2
CH
3
O
















