
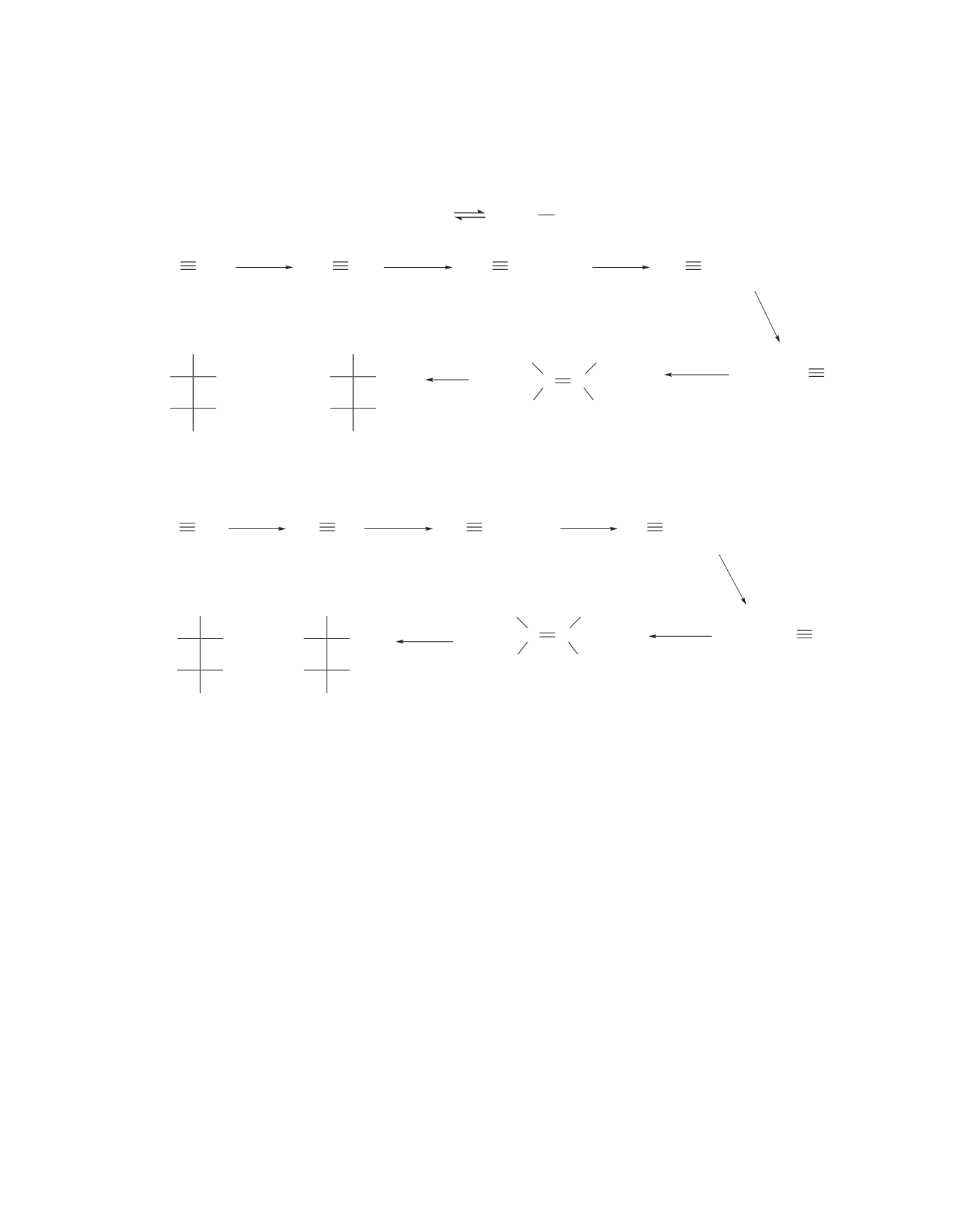
256 Chapter 7
Copyright © 2017 Pearson Education, Inc.
56.
If HO
-
and HOOH are added at the same time as BH
3
1
or R
2
BH
2
, HO
-
is a better nucleophile than the
alkyne (no bonds have to be broken when HO
-
reacts with the electrophile), so BH
3
1
or R
2
BH
2
will react
with HO
-
instead of with the alkyne.
+ −
HO
HO
−
BH
3
+
BH
3
57.
a.
NaNH
2
NaNH
2
HC CH
HC C
CH
3
CH
2
Br
HC CCH
2
CH
3
Na/NH
3
(liq)
–78 ˚C
C CCH
2
CH
3
CH
3
CH
2
Br
–
CCH
2
CH
3
–
CH
2
CH
3
H OH
CH
2
CH
3
H Br
CH
2
CH
3
HO H
CH
2
CH
3
Br
H
Br
2
H
2
O
+
(3
S
,4
R
)-4-bromo-
3-hexanol
(3
R
,4
S
)-4-bromo-
3-hexanol
H
C C
CH
2
CH
3
CH
3
CH
2
H
CH
3
CH C
2
b.
NaNH
2
NaNH
2
HC CH
HC C
CH
3
CH
2
Br
HC CCH
2
CH
3
C CCH
2
CH
3
CH
3
CH
2
Br
Br
2
H
2
O
CH
3
CH
2
C C
CH
2
CH
3
H
H
H
2
Lindlar
catalyst
CH
3
CH
2
C CCH
2
CH
3
–
–
+
(3
R
,4
R
)-4-bromo-
3-hexanol
(3
S
,4
S
)-4-bromo-
3-hexanol
CH
2
CH
3
HO H
CH
2
CH
3
H Br
CH
2
CH
3
H OH
CH
2
CH
3
Br
H
Because anti addition occurs in the last step, the threo enantiomers are formed.
If Na
>
NH
3
1
liq
2>
-
78
°
C is used instead of H
2
>
Lindlar catalyst in the fifth step, the trans alkene will be
formed. Reaction of Br
2
and H
2
O with the trans alkene will form the erythro enantiomers.
58.
(3
E
,6
E
)-3,7,11-trimethyl-1,3,6,10-dodecatetraene
The configuration of the double bond at the 1-position and at the 10-position is not specified because
isomers are not possible at those positions, because there are two hydrogens bonded to C-1 and two methyl
groups bonded to C-11.
















